Discover Chlorophyll Variety in Different Plants Using Paper Chromatography
Abstract
Do you like to analyze stuff and finding out what it is made of? An analytical chemistry method, called chromatography, allows you to separate mixtures of compounds and to identify each individual compound within the mixture. Chromatography is used by many scientists, for example food scientists, forensic scientists, or organic scientists to analyze all kinds of mixtures such as food, blood, or medicine. In this project, you will be using paper chromatography to analyze the pigments from different plant leaves. What do you think you will find?
Summary
None
A kit is available from our partner Home Science Tools®. See the Materials section for details.
No issues
Author: Amber Hess
Editor: Andrew Olson, Science Buddies
Edited by Svenja Lohner, PhD, Science Buddies
Objective
The objective of this project is to use paper chromatography to analyze the leaf pigments found in three different types of plants.
Introduction
samples of green leaves of plants for discovering chlorophyll
Plants
Plants need to absorb light in order to create their own food in form of glucose. They do this with the help of a special pigment mixture located in their leaves, which include chlorophyll a, chlorophyll b, and carotenoids. Chlorophyll absorbs photons, which excite electrons in its central magnesium (Mg) atom. These electrons are channeled away from the chlorophyll to be used as energy to create glucose. Figure 1 shows the chemical structure for chlorophyll. Chlorophyll a has one carbon and three hydrogen atoms in place of the "R" group, and chlorophyll b has a carbon, a hydrogen, and an oxygen atom. The red highlighted part of the image is called the porphyrin ring. Each "point" in the diagram (i.e. each tip of the pentagons, hexagons, and jagged edges) represents a carbon atom. Hydrogen atoms are attached to most of the carbon atoms, but they are not shown in this structure.

Chlorophyll comes in two forms depending on their chemical structure, but both forms contain a porphyrin ring. A porphyrin ring has a magnesium atom at its center and is surrounded by nitrogen atoms.
Figure 1. The molcular structure of chlorophyll includes a central magnesium atom and a porphyrin ring (highlighted in red).
Due to its specific molecular structure, chlorophyll is able to absorb red and blue light from the sun, but it does not absorb green light, it reflects it. This is why tree leaves are normally green. In the fall, the tree shuts down chlorophyll production to save energy, and recycles it for later use. Thus the green-reflecting chlorophyll disappears, and we are left with the orange and red carotenoids (pigments that capture light energy and transfer it to chlorophyll), and so the leaves turn red and orange.
Chromatography
Before you continue reading, watch the video. The video gives an overview of what paper chromatography is, shows how it is done, explains the separation processes involved, and also provides tips and tricks for troubleshooting your experiment.
In this project, you will be using paper chromatography to investigate chlorophyll and other pigments in plant leaves. Paper chromatography is a technique, that is used to separate the various components in a complex mixture or solution. In each chromatography apparatus there is generally a mobile phase, which is a fluid that runs along the stationary phase, and a stationary phase, that stays stationary while the mobile phase moves through. You will be extracting chlorophyll and other dye pigments from plant leaves. Then you will make a small spot of this extract onto a strip of chromatography paper. The bottom of this strip will be placed in a beaker of acetone, and the acetone will soak up into the paper. The acetone (solvent) is the mobile phase of the chromatography system, whereas the paper is the stationary phase. These two phases are the basic principles of chromatography. Chromatography works by something called capillary action. The attraction of the acetone to the paper (adhesion force) is larger than the attraction of the acetone to itself (cohesion force), hence the acetone moves up the paper. The chlorophyll and other plant pigments will also be attracted to the paper, to themselves, and to the acetone differently, and thus a different component will move a different distance depending upon the strength of attraction to each of these objects, as shown in Figure 2. As an analogy, let's pretend you are at a family reunion. You enjoy giving people hugs and talking with your relatives, but your cousin does not. As you make your way to the door to leave, you give a hug to every one of your relatives, and your cousin just says "bye." So, your cousin will make it to the door more quickly than you will. You are more attracted to your relatives, just as some chemical samples may be more attracted to the paper than the solvent, and thus will not move up the solid phase as quickly. Your cousin is more attracted to the idea of leaving, which is like the solvent (the mobile phase).

A homemade paper chromatography testing box is made from a tall box with a lid. A dowel spanning the width of the box is placed near the top to allow a binder clip to hold a paper strip that has been marked by colored pigments. The paper strip is long enough to reach the bottom of the box where there is a small pool of solvent. As the solvent is absorbed by the paper and moves upward it brings some of the colored pigment markings with it.
Figure 2. Paper chromatography. Molecules are separated from each other, depending on how fast they migrate with the solvent up the chromatography paper. (Wikipedia, 2008.)
In paper chromatography, you can see the components separate out on the chromatography paper and identify the components based on how far they travel. To do this, we calculate the retention factor (Rf value) of each component. The Rf value is the ratio between how far a component travels and the distance the solvent (mobile phase) travels from a common starting point (the origin). For example, if one of the sample components moves 2.5 centimeters (cm) up the paper and the solvent moves 5.0 cm, as shown in Figure 3 below, then the Rf value is 0.5. You can use Rf values to identify different components as long as the solvent, temperature, pH, and type of paper remain the same. In Figure 3, the light blue shading represents the solvent and the dark blue spot is the colored solution sample.

The distance from the top of the blue pigment to the origin line is divided by the distance of the solvent front to the origin line to solve for an Rf value.
Figure 3. For each compound, an Rf value is calculated based on how far it traveled along the stationary phase. In paper chromatography, Rf values are used to compare different components to each other.
Rf values are calculated by looking at the distance each component travels on the chromatography paper compared to the distance traveled by the solvent front. This ratio will be different for each component due to its unique chemical properties.
When measuring the distance the component traveled, you should measure from the origin (where the middle of the spot originally was) and then to the center of the spot in its new location. To calculate the Rf value, we then use Equation 1 below.
Equation 1:
In our example, this would be:
Note that an Rf value has no units because the units of distance cancel.
Polarity
Polarity has a huge affect on how attracted a chemical is to other substances. Some molecules have a positively charged side and a negatively charged side, similar to a magnet. The positive side is attracted to the negative side of another molecule (opposites attract), and vice versa. The larger the charge difference, the more polar a molecule is. The reason for the unequal charge is that electrons (which are negatively charged) are not shared equally by each atom (in water, the negative electrons are more attracted to the oxygen because of its atomic structure). Some molecules, like vegetable oil, are neutral and do not have a charge associated with them; they are called nonpolar molecules. Polarity affects many of a molecule's properties, such as its affinity to water. Water is a highly polar molecule, so other polar molecules are easily attracted to it. A molecule is called hydrophilic if it dissolves well in water (hydrophilic essentially means "loves water"). A nonpolar molecule, such as oil, does not dissolve well in water, and thus it is hydrophobic ("fears water"). Oil would rather stick to itself than to water, and this is why it forms a layer across water instead of mixing with it. In chromatography, the polarity of the solvent determines how strongly the individual components of the mixture are attracted to the mobile phase versus the stationary phase. Therefore, the separation process will be dependent on the polarity of both phases.
Figure 4 below shows a water molecule bonding with another water molecule. The negatively charged oxygen atom (red) is attracted to the positively charged hydrogen atom (white) on the other molecule.
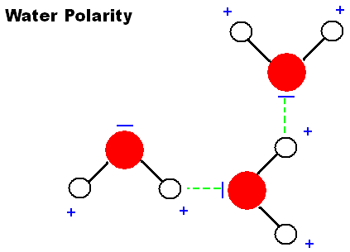
Molecules of water are made of a single oxygen atom and two hydrogen atoms. Oxygen atoms have a negative charge and hydrogen atoms have a positive charge. Hydrogen bonds can form between the hydrogen and oxygen atoms of different water molecules.
Figure 4. Hydrogen bond formation between two adjacent water molecules.
Figure 5 shows a fatty acid (a component of fat molecules) bonding with water. The hydrophobic tail is not attracted to the water, and thus it stands upright out of the water. The hydrophilic head is attracted to the water, which bonds to it. Many fatty acid molecules bonded together can form a layer above the water. Carbon atoms are represented by black circles.

The head of a fatty acid molecule can create a hydrogen bond with water molecules. Hydrogen can bond to carbon atoms in the fatty acid and subsequent oxygen atoms in water can bond to the hydrogen attached to the carbon.
Figure 5. Hydrogen bond formation between a fatty acid molecule and a water molecule.
Chromatography Applications
Chromatography is used in many different industries and labs. The police and other investigators use chromatography to identify clues at a crime scene like blood, ink, or drugs. More accurate chromatography in combination with expensive equipment is used to make sure a food company's processes are working correctly and they are creating the right product. This type of chromatography works the same way as regular chromatography, but a scanner system in conjunction with a computer can be used to identify the different chemicals and their amounts. Chemists use chromatography in labs to track the progress of a reaction. By looking at the sample spots on the chromatography plate, they can easily find out when the products start to form and when the reactants have been used up (i.e., when the reaction is complete). Chemists and biologists also use chromatography to identify the compounds present in a sample, such as plants — exactly as you are doing in this project!
In this science project, you will be extracting color pigments, including chlorophyll, from different plants, and analyze the extracts using paper chromatography. Which types of chlorophyll and other pigments do you think you will find?
Terms and Concepts
- Carotenoid
- Chlorophyll
- Paper chromatography
- Mobile phase
- Stationary phase
- Capillary action
- Adhesion force
- Cohesion force
- Rf value
- Polarity
- Hydrophilic
- Hydrophobic
Questions
- Why do trees leaves turn different colors in the fall?
- What are the different pigments used for in the plant?
- What is the process of photosynthesis?
- What wavelengths of light do plants use?
- What makes a molecule polar or nonpolar? How does its polarity affect its interactions with other molecules?
- How do the polarities of the solid phase and liquid phase affect how far chemicals travel on the paper strip/plate?
Bibliography
Here are some basic chemistry and chromatography resources:
- Andrew Radar Studios staff. (n.d.) Chem4KIDS. Retrieved March 18, 2013.
- Carpi, Anthony, Ph.D. (2003). Chemical Bonding. Visionlearning. Vol CHE-1 (7). Retrieved March 18, 2013.
- Clark, Jim. (2007). Paper Chromatography. Retrieved March 18, 2013.
These resources will give you more information about chromatography and teach you about the types of chromatography used in research labs today:
- Science Buddies. (n.d.). Paper Chromatography Resources. Retrieved January 14, 2018.
- Waters Corporation Staff. (2012). High Performance Liquid Chromatography. Retrieved November 29, 2012.
You can start learning more about plants and pigments from these resources:
- Davidson, Michael W. (n.d.). Plant Cell Structure. Molecular Expressions. Retrieved March 18, 2013.
- Wikipedia Contributors. (2013, March 15). Biological Pigment. Wikipedia, The Free Encyclopedia. Retrieved March 18, 2013.
- ProFlowers. (2015, May 22). How Flowers Get Their Color. Retrieved June 30, 2020.
Materials and Equipment 
Recommended Project Supplies
- Candy Chromatography Science Kit, available from our partner Home Science Tools®. You will need these items from the kit:
- Chromatography paper strips; 9. The kit comes with 20 strips; additional chromatography paper can be purchased separately from our partner Home Science Tools®.
- 100 mL beaker
- Mini binder clips (2)
- Wooden splints
- Pipette
- Note: This kit contains additional items to do other chromatography science projects. See the kit instructions page for details. Downloadable materials safety data sheets (MSDS) are available for all kit chemicals if required by your science fair.
- You will also need to gather these items, not included in the kit:
- Various plant leaves:
- Spinach leaves
- Iceberg lettuce leaves
- Marigold leaves
- Mortar and pestle
- Test tubes (9)
- Acetone (nail-polish remover)
- Water
- Ruler
- Pencils
- Lab notebook
- Various plant leaves:
Disclaimer: Science Buddies participates in affiliate programs with Home Science Tools®, Amazon.com, Carolina Biological, and Jameco Electronics. Proceeds from the affiliate programs help support Science Buddies, a 501(c)(3) public charity, and keep our resources free for everyone. Our top priority is student learning. If you have any comments (positive or negative) related to purchases you've made for science projects from recommendations on our site, please let us know. Write to us at [email protected].
Experimental Procedure

- Grind up roughly equal samples of each of the different plant leaves and distribute them into test tubes. There should be at least three labeled test tubes for each type of plant (if using the Iceberg lettuce, "Iceberg 1," "Iceberg 2," and "Iceberg 3" are good names for the tubes).
- Add enough acetone (nail-polish remover) to suspend the ground-up leaves.
- Let the acetone/leaf mixture sit for 24 hours.
- Cut each chromatography paper in half (length-wise) to make approximately 2 centimeters (cm) wide by 7.5 cm long strips. You will need at least 9 chromatography strips.
- Use a pencil to lightly label which leaf extract will be spotted on each paper strip. Label three chromatography strips for each type of plant leave. Tip: do not use a pen for writing on the strips: the ink will run when the solvent passes through the strips.
- Draw a pencil line 1 cm from the edge of each strip of paper, as shown in Figure 6 below.
- This will be the origin line or baseline.
- You will spot the plant extract for each strip right on this line, as shown in Figure 6.
- Take a pipette and fill it with one of the plant leaf extracts.
- Spot the sample in the middle of the origin line (see Figure 6, below). Let the sample air dry. Then apply another layer of the sample on the same spot. Ideally, you want to repeat this step 5–10 times to increase the concentration of your sample on the paper strip.
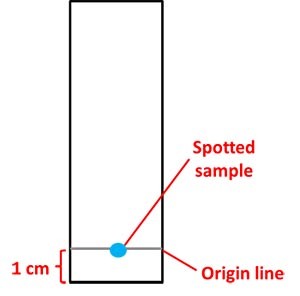 Figure 6. Each chromatography strip will have an origin line (baseline). The plant extract to be tested will be spotted in the middle of the origin line.
Figure 6. Each chromatography strip will have an origin line (baseline). The plant extract to be tested will be spotted in the middle of the origin line. - Repeat step 8 with the other plant leaf extracts you want to test. Use a new paper strip for each one.
- Fill the 100 mL beaker to a depth of 1 cm with the acetone (nail-polish remover).
- Clip two of the prepared chromatography strips to a wooden splint. Make sure the two strips do not touch each other or the beaker and that their bottoms are aligned. Rest the splint on top of the beaker so that the strips hang straight into the beaker.
- If necessary, add more of the acetone. The goal is to have the end of the chromatography strip just touching the surface of the solvent solution, as shown in Figure 7 below.

Figure 7. Your setup should look similar to this example. The end of the chromatography strip should just touch the acetone. Note: This picture does not show chromatography strips with plant extract. The colors on your paper strips should look different.
- Let the solvent rise up the strip (by capillary action) until it is about 0.5 cm from the top then remove the strip from the solvent. Depending on the type of chromatography paper and solvent, the separation process can take anywhere from 30 minutes to several hours. Regularly check on your chromatography strip and the solvent front — if you let it run too long the plant extract may run off the paper and become distorted.
- Use a pencil to mark how far the solvent rose.
- Allow the chromatography strip to dry, then measure (in centimeters) and calculate the Rf value for each plant pigment. Record your results in your lab notebook.
- Tip: Use Equation 1, which is given in the Introduction, for calculating the Rf value.
- Repeat this experiment for each of your plant extracts three times.
Questions
- What types of pigments do you think are present in each type of leaf? How can you tell? (Consider color, Rf value, etc.)
- How were the Rf values different for each pigment and leaf? Why? (Make sure to consider molecular structure, polarity, etc.)
- How do the different pigments help the plant? Why do different plants have different amounts and different types of these pigments?
Ask an Expert
Variations
- Do plants' pigments vary with more or different types of light? You might try growing a few plants of the same type under different colored lights, and/or more intense light, and then do the chromatography procedure. If you are very precise when taking samples (i.e., measuring how much of the leaf you grind up for each plant), you could even comment how the amount of each pigment changes, if at all.
- You can also analyze the amino acids that are found in orange juice and lemon juice with chromatography.
- For a less advanced chromatography project, see Explore How Chromatography Can Unmix Mixtures
Careers
If you like this project, you might enjoy exploring these related careers:
Contact Us
Our kits are developed in partnership with Home Science Tools®. If you have purchased a kit for this project, Home Science Tools® is pleased to answer any questions.In your email, please follow these instructions:
- Include your Home Science Tools® order number.
- Please describe how you need help as thoroughly as possible:
Examples
Good Question I'm trying to do Experimental Procedure step #5, "Scrape the insulation from the wire. . ." How do I know when I've scraped enough?
Good Question I'm at Experimental Procedure step #7, "Move the magnet back and forth . . ." and the LED is not lighting up.
Bad Question I don't understand the instructions. Help!
Good Question I am purchasing my materials. Can I substitute a 1N34 diode for the 1N25 diode called for in the material list?
Bad Question Can I use a different part?
Contact Support