Abstract
Cooking, washing, and many industrial processes involve the mixing of liquids. How liquids behave when they are mixed is dependent on many factors. Sometimes, the mixing behavior of liquids can be quite surprising! For example, when you mix 50 mL of one liquid with 50 mL of another liquid, you might not end up with a 100 mL mixture! In this science project, you will explore this phenomenon, which we call the "excess volume" of mixtures. How do you think your mixing volumes will change when you make solutions of different ratios of water and rubbing alcohol?Summary
Objective
Investigate the excess volume of water and isopropyl alcohol solutions with different mixing ratios.
Introduction
Mixing liquids is something we do every day (Figure 1). Every time we make a beverage with different liquids, like coffee and milk, we mix liquids. During cooking or baking, we often mix liquids in order to make things like sauces, soups, or cake batter. And when we wash laundry with detergent or wash our hair with shampoo, we mix these with water, which is also a liquid!
While we usually do not think much about what happens when we mix different liquids, it is an important consideration for many applications. For industrial processes like distillation or polymerization, for example, the mixing parameters must be carefully controlled to achieve the desired product. In environmental science, it is important to know how liquids such as pollutants mix with water. Knowing their mixing behaviors allows scientists to determine how pollutants spread and to find the best clean-up strategies. Studying the mixing behavior of liquids can help people understand and optimize a wide range of chemical and physical processes in industry and in the environment.

Figure 1. Making beverages, cooking or baking, and washing clothes are all everyday activities that require the mixing of liquids.
So, what happens when different liquids are mixed? The answer is—it depends! Sometimes, mixing different liquids can result in a chemical reaction. But even if liquids do not react with each other, their mixing behaviors can vary. How substances combine, or mix, is defined by their miscibility. Some liquids mix to form something called a solution. Solutions are homogeneous mixture, which means that you cannot distinguish the two liquids anymore. In contrast, when liquids cannot be mixed together (they are "immiscible"), they form separate layers. The result is a heterogeneous mixture.
Whether a liquid mixes with another is dependent on the liquids' individual molecular structures. Molecules can be classified into polar and nonpolar molecules. When atoms come together to form a molecule, they share negatively charged electrons in a chemical bond. Sometimes, one atom attracts the electrons more than the other atom does, which results in a slight separation of the charge into a positive and negative pole within the molecule. This is called an electric dipole. When this happens, the molecule is usually a polar molecule. Molecules that have an equal charge balance are nonpolar molecules (Figure 2).

Left: A schematic diagram of a water molecule, which is an example of a polar molecule. Two blue circles represent the hydrogen atoms and a red circle represents the oxygen atom. Grey lines connecting the circles represent the chemical bonds between the atoms. A plus sign next to each hydrogen atom and a minus sign next to the oxygen atom show the charge imbalance within the molecule. Right: A schematic diagram of a carbon dioxide molecule, which is an example of a nonpolar molecule. Two red circles represent the oxygen atoms and a black circle represents the carbon atom. Grey lines connecting the circles represent the chemical bonds between the atoms.
Figure 2. The water molecule (left) is made from one oxygen (O) atom and two hydrogen (H) atoms. Molecules that have a charge imbalance, like water, are polar. The carbon dioxide molecule (right) is made from one carbon (C) and two oxygen (O) atoms. Molecules that have an equal charge balance, like carbon dioxide, are nonpolar.
Once molecules are in a solution, they can interact with each other. For example, molecules can attract each other or repel each other, depending on their charges. These interactions create intermolecular forces in solutions. One example of an intermolecular force is the formation of hydrogen bonds between molecules. These bonds are caused by the attraction between individual molecules. Molecules that are polar tend to form hydrogen bonds, whereas nonpolar molecules do not tend to form hydrogen bonds. Figure 3 shows water molecules bonding with one another (hydrogen bonding). The negatively charged oxygen atom on one water molecule is attracted to the positively charged hydrogen atom on another water molecule.
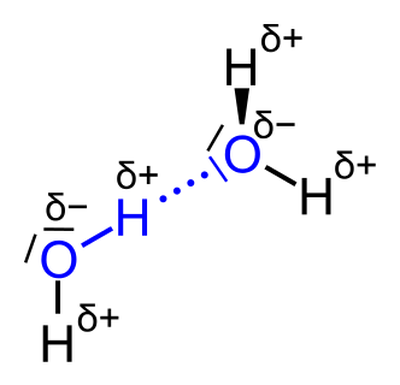
Each water molecule is represented by an O that is connected by lines to two Hs. The lines between the oxygen and hydrogen atoms represent the chemical bonds within the molecule. A blue dotted line connects the oxygen atom of one water molecule with a hydrogen atom of the other water molecule; this represents a hydrogen bond. There are also delta plus and delta minus signs next to the atoms, which represent positive and negative charges.
Figure 3. Polar molecules, such as water, tend to form hydrogen bonds, which are one type of intermolecular force.
Intermolecular forces can result in unusual properties within a solution. These are called excess properties. Excess properties of a mixture define the difference between the actual property of that mixture and its expected property.
One example of an excess property is excess volume. If you added 50 mL of one liquid to 50 mL of another liquid, you would expect the resulting mixture to have a volume of 100 mL. However, this is not always the case. Sometimes the volume of the mixture adds up to more than or less than 100 mL. This difference in volume is called the "excess volume." The excess volume can be positive or negative. When the actual volume of the mixture is less than the sum of the volumes of the original liquids, a volume contraction has happened, and the excess volume is negative. Why does this happen?
There are several reasons for the volume contraction. Hydrogen bonding between the different molecules, as mentioned above, often leads to a more compact structure of the mixture compared to the individual liquids. Other factors are molecule size and molecule shape. Every molecule has a different size and shape. If liquids with large, bulky molecules are mixed with a liquid with small molecules, the small molecules can slide into the spaces between the large molecules, leading to a compact packing structure of the mixture. Imagine that the large molecules are golf balls in a bucket. To the golf balls, you add a bucket of sand, which represents very small molecules. The sand would not just sit on top of the golf balls, but would flow down into the spaces between the golf balls. The volume of the golf balls and sand combined will be less than their individual volumes added together. This is exactly what happens with the large and small molecules in mixtures.
In this science project, you will investigate how the mixing ratio of two different liquids affects the excess volume of the mixture. We will use the mole fraction as a way to measure how much of one type of molecule there is in the mixture. The mole fraction is defined as the ratio of the number of moles of a particular substance to the total number of moles of all the substances in a mixture (Equation 1).
Equation 1:where
= mole fraction of the substance "i"= number of moles of the substance "i"
= total number of moles of all substances in the mixture
The mole fraction is a useful way to express the composition of mixtures in a solution. It helps scientists understand how different molecules in a mixture interact with each other, and it allows them to predict how solutions will behave under different conditions.
The two liquids you will use to make your mixture in this experiment are water and isopropyl alcohol. There is just one thing left to say before you start with your experiment: Be very precise with your volume measurements, as every milliliter counts!
Terms and Concepts
- Miscibility
- Solution
- Homogeneous
- Heterogeneous
- Polar
- Non-Polar
- Electrons
- Electric dipole
- Intermolecular force
- Hydrogen bond
- Excess property
- Excess volume
- Volume contraction
- Mole fraction
- Isopropyl alcohol
Questions
- Why is the mixing behavior of liquids relevant?
- What is excess volume, and why does it occur?
- What are examples of mixtures with volume contraction?
- What factors affect the excess volume of mixtures?
Bibliography
- Dortmund Data Bank (DDB). (n.d.). Ve - Excess Volumes, Mixture Densities. Retrieved May 18, 2023.
- Dortmund Data Bank (DDB). (n.d.). Excess Volume Data. Retrieved May 18, 2023.
Materials and Equipment
- Measuring cylinders, 100 mL (2), Note: You should use two identical measuring cylinders (same brand), as different ones can be calibrated differently.
- Large plastic cups (2)
- Permanent marker
- Nitrile gloves
- Safety goggles
- Isopropyl alcohol, 99% (about 900 mL)
- Water
- Optional: Two different colors of food coloring
- Cloth or paper towels
- Wooden skewers (2)
- Digital Scale, 0.1 g precision
- Calculator
- Empty, sealable 2 L bottle or container to store the isopropyl alcohol waste
- Pen or pencil
- Lab notebook
Disclaimer: Science Buddies participates in affiliate programs with Home Science Tools®, Amazon.com, Carolina Biological, and Jameco Electronics. Proceeds from the affiliate programs help support Science Buddies, a 501(c)(3) public charity, and keep our resources free for everyone. Our top priority is student learning. If you have any comments (positive or negative) related to purchases you've made for science projects from recommendations on our site, please let us know. Write to us at [email protected].
Experimental Procedure

Preparing the Experiment
- Label the two plastic cups "water" and "isopropyl alcohol."
- Label the two transfer pipettes "water" and "isopropyl alcohol."
- Label the empty 2 L bottle "isopropyl alcohol waste."
- Fill the water cup with tap water. Fill the isopropyl alcohol cup with 99% isopropyl alcohol. Let both liquids adjust to room temperature. Note: When handling the isopropyl alcohol, always wear gloves and safety goggles!
- Optional: Add one drop of food coloring to each cup. Each liquid should have a different color. This makes it easier to keep track of them during the experiment. The color will also make it easier to read the liquid volume in the measuring cylinder.
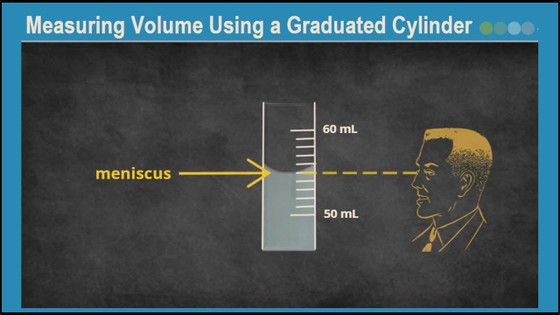
- Practice reading liquid volumes in the measuring cylinder.
- Solutions in narrow containers, such as measuring cylinders, tend to creep up the sides of the walls at the surface of the liquid. This is due to the surface tension of water. The surface of the liquid thus forms a curve called a meniscus. To measure the volume of the liquid in the measuring cylinder, always read the point on the graduated scale that coincides with the bottom of the meniscus.
- You will need to view the meniscus at eye level. Reading the meniscus at an angle other than eye level will give inaccurate results. Watch the video below for an example of reading volume in a measuring cylinder.
- Fill the cylinder with a random amount of water from your water cup and practice reading the water volume. Repeat the process several times. Pour the water back into the cup in between to reuse it.
- Check whether your two 100 mL measuring cylinders are calibrated similarly, meaning they both show the same volume for the same amount of water.
- Pour 50 mL water into one of the measuring cylinders. Use the "water" transfer pipette for the final milliliters to fill the cylinder up to the 50 mL mark. Be precise! The bottom of the meniscus should line up with the 50 mark when viewed at eye level!
- Pour the 50 mL water from this measuring cylinder into the second measuring cylinder. Gently tap or shake the cylinder to get out any remaining drops that are stuck to the inside.
- Read the water volume in the second measuring cylinder. It should also read 50 mL. There might have been minimal loss due to the liquid transfer, but not more than 0.5 mL.
- If the water volume in the two cylinders differs by more than 1 mL, you will not be able to use them for this experiment. Try to find two measuring cylinders that have a better calibration match.
Measuring the Excess Volume
You will be preparing different mixtures of water and 99% isopropyl alcohol. The different mixing ratios are given in Table 1. The table also states the mole fraction of isopropyl alcohol for each mixture. How the mole fraction was calculated is explained in the Technical Note below.
- With a permanent marker, label the measuring cylinders "water" and "isopropyl alcohol."
- Measure the mass of both of your measuring cylinders. You will need these values for calculating the mass of your liquid mixtures later.
- Switch the scale on and zero it.
- Place one of the empty cylinders on the scale.
- Record its mass in your lab notebook.
- Repeat steps a–c with the second measuring cylinder.
| Mixture # | Volume water [mL] | Volume isopropyl alcohol [mL] | Mole fraction isopropyl alcohol (xisopropyl alcohol) | Mass water measured [g] | Mass isopropyl alcohol measured [g] | Mass mixture measured (water + isopropyl alcohol in measuring cylinder) [g] | Volume mixture measured [mL] (water + isopropyl alcohol) |
|---|---|---|---|---|---|---|---|
| 1 | 5 | 50 | 0.7 | ||||
| 2 | 10 | 50 | 0.54 | ||||
| 3 | 30 | 50 | 0.28 | ||||
| 4 | 50 | 50 | 0.19 | ||||
| 5 | 50 | 30 | 0.12 | ||||
| 6 | 50 | 10 | 0.045 | ||||
| 7 | 25 + 25 | 0 | 0 | ||||
| 8 | 0 | 25 + 25 | 1 |
- Start with mixture 1 as shown in row 1 in Table 1.
- Place the "water" measuring cylinder on the scale and zero it.
- Add 5 mL of water to the cylinder. Use the "water" transfer pipette to fill the cylinder precisely to the 5 mL mark.
- Record the mass (to the first decimal) of the water in your data table.
- Place the "isopropyl alcohol" measuring cylinder on the scale and zero it.
- Add 50 mL of isopropyl alcohol to the cylinder. Use the "isopropyl alcohol" transfer pipette to fill the cylinder precisely to the 50 mL mark.
- Record the mass (to the first decimal) of the isopropyl alcohol in your data table.
- Carefully pour the contents of the "water" cylinder into the "isopropyl alcohol" cylinder on the scale. Try to transfer every single drop of water from one cylinder to the other.
- Record the mass (to the first decimal) of the mixture in your data table.
- With a wooden skewer, mix the contents in the measuring cylinder.
- Wait until the bubbles have subsided, then read the volume of the mixture in the measuring cylinder. Record the volume to the first decimal in your data table.
- Discard the mixture into the prepared 2 L waste bottle.
- Briefly rinse the two measuring cylinders and dry them with a cloth or paper towels. You can use another wooden skewer to push the cloth all the way to the bottom of the graduated cylinder. Both cylinders need to be dry before continuing with the next step.
- Repeat steps a–l two more times, so you have measurements from three independent trials for each mixture. You will need to create a new data table for each trial.
- Repeat step 3 for mixtures 2–6 listed in Table 1.
- The last two measurements will be mixtures of water with water and isopropyl alcohol with isopropyl alcohol. These measurements correspond to an isopropyl alcohol mole fraction of 0 and 1.
- Repeat step 3, but instead of measuring water in one measuring cylinder and isopropyl alcohol in the second, you will be adding 25 mL of water to each cylinder. This measurement corresponds to an isopropyl alcohol mole fraction of 0.
- Repeat step 3, but instead of measuring water in one measuring cylinder and isopropyl alcohol in the second, you will be adding 25 mL of isopropyl alcohol to each cylinder. This measurement corresponds to an isopropyl alcohol mole fraction of 1.
- You will need to dispose of your isopropyl alcohol waste at your local hazardous waste drop-off location. You should not dispose of the alcohol down the sink or toilet—that is only safe with isopropyl alcohol solutions containing less than 5% of isopropyl alcohol. The solutions in this experiment are much stronger.
Analyzing your data
- Once you have done three measurements for each mixture listed in Table 1, you are ready to analyze your data. You are going to compare the theoretical mass and volume of the mixtures (the sums of the two original amounts) with the actual measurements you obtained.
- Start by calculating the average mass of each of your mixtures from all three trials.
- To get the average mass of the mixture, add the total mass of the mixture for each trial and divide the result by three.
- From the calculated average mass, subtract the mass of the empty measuring cylinder you wrote down earlier in your lab notebook. You want to know the mass of just the liquid mixture.
- Record your result in a data table like Table 2.
- Do these calculations for each mixture.
| Mixture # | Volume water [mL] | Volume isopropyl alcohol [mL] | Mole fraction isopropyl alcohol (xisopropyl alcohol) | Theoretical mass of mixture [g] | Average mass of mixture measured [g] | Theoretical volume of mixture [mL] | Average volume of mixture measured [mL] |
|---|---|---|---|---|---|---|---|
| 1 | 5 | 50 | 0.7 | 44.24 | |||
| 2 | 10 | 50 | 0.54 | 49.23 | |||
| 3 | 30 | 50 | 0.28 | 69.19 | |||
| 4 | 50 | 50 | 0.19 | 89.15 | |||
| 5 | 50 | 30 | 0.12 | 73.45 | |||
| 6 | 50 | 10 | 0.045 | 57.75 | |||
| 7 | 25 + 25 | 0 | 0 | 49.9 | |||
| 8 | 0 | 25 + 25 | 1 | 39.25 |
- Calculate the average measured volume of each of your mixtures from all three trials.
- To get the average volume of the mixture, add the measured volumes of the mixture for each trial and divide the result by three.
- Record your result in your data table.
- Do these calculations for each mixture.
- The theoretical mass of each of your mixtures is given in Table 2. You will find information on how to calculate the combined mass of the mixture in the Technical Note below.
- Calculate the theoretical volume of each mixture.
- Add together the original volume of water and the original volume of isopropyl alcohol that you mixed together for each mixture.
- Record the results in your data table.
- Calculate the difference between the theoretical and the measured mass average for each mixture. Record your results in a data table like Table 3.
- Subtract the measured average mass from the theoretical mass for each mixture.
- Record your results in your data table.
- Note that the result can be either a positive or a negative number. If the result is negative, the mass of the mixture has decreased; if the result is positive, the mass of the mixture has increased.
| Mole fraction isopropyl alcohol (xisopropyl alcohol) |
Total moles of mixture [mol] | Mass difference [g] | Volume difference (Excess Volume) [mL] | Excess Volume [mL/mol] |
|---|---|---|---|---|
| 0.7 | 0.93 | |||
| 0.54 | 1.2 | |||
| 0.28 | 2.31 | |||
| 0.19 | 3.42 | |||
| 0.12 | 3.162 | |||
| 0.045 | 2.9 | |||
| 0 | 2.85 | |||
| 1 | 0.65 |
- Calculate the difference between the theoretical and the measured volume average for each mixture. This number represents the excess volume of your mixture.
- Subtract the measured average volume from the theoretical volume for each mixture.
- Record your results in your data table.
- Note that the result can be either a positive or a negative number. If the result is negative, the volume of the mixture has decreased; if the result is positive, the volume of the mixture has increased.
- The excess volume is often recorded in milliliters per mole. The use of volume per mole as a unit allows for a standardized comparison of excess volume across different mixtures and concentrations. Divide the calculated volume difference by the total moles of the mixture, which are given in Table 3. You will find information on how to calculate the total moles of the mixture in the Technical Note below.
- Now it is time to visualize your data. Make a bar graph that shows the theoretical and the measured mass of the mixtures side by side.
- In your graph, put the mole fraction of isopropyl alcohol on the x-axis.
- Add two data series to the graph: one showing the measured average mass of the mixture, and one showing the theoretical mass of the mixture.
- Make another bar graph that shows the theoretical and the measured volume of the mixtures side by side.
- In your graph, put the mole fraction of isopropyl alcohol on the x-axis.
- Add two data series to the graph: one showing the measured average volume (excess volume) of the mixture, and one showing the theoretical volume of the mixture.
- To be able to compare your measurements to measurements with other mixtures or
measurements made by someone else, create one more graph that shows how the excess volume changes depending on the mole fraction of isopropyl alcohol on a per-mole basis.
- Make a scatter plot of your data.
- In your scatter plot, put the mole fraction of isopropyl alcohol on the x-axis.
- Put the excess volume in mL/mol on the y-axis.
- Finally, use all your data and graphs to answer the following questions.
- How did the measured volumes of your mixtures deviate from the expected volume? Did you expect these results?
- How did the measured masses of your mixtures deviate from the expected volume? Did you expect these results?
- Did the volume and mass of the mixtures behave differently? If yes, can you explain your results?
- What relationship did you find between the excess volume and the mixing ratio of the two liquids (mole fraction of isopropyl alcohol)?
In Table 1, you will find the isopropyl alcohol mole fractions for each mixing ratio. Below we will explain how to calculate these mole fractions. The following calculations will also show how to calculate the theoretical mass of the mixture, given in Table 2, and the total moles of the mixture, given in Table 3.
To calculate the mole fraction of isopropyl alcohol, we need to know the density and molar mass of water and isopropyl alcohol, which are both parts of the liquid mixture. The information is given below.
Density at 20°C
ρH2O = 0.998 g/mLρisopropyl alcohol = 0.785 g/mL
Molar mass
MH2O = 18 g/molMisopropyl alcohol = 60 g/mol
The values above are for 100% isopropyl alcohol. They will not differ significantly for 99% isopropyl alcohol, which we use in this experiment.
In the following example, we will calculate the mole fraction of a mixture with 10 mL of water and 50 mL of isopropyl alcohol.
In the first step, we will use Equation 2 to calculate the mass of both liquids.
Equation 2:m = mass of substance [g]
V = volume of substance [mL]
Using Equation 2, the mass of 10 mL water is:
m = ρ x V = 0.998 g/mL × 10 mL = 9.98 gSimilarly, the mass of 50 mL isopropyl alcohol is:
m = ρ x V = 0.785 g/mL × 50 mL = 39.25 gThe combined mass of both solutions is:
mtotal = mH2O + misopropyl alcohol = 9.98 g + 39.25 g = 49.23 gIn the next step, we will use Equation 3 to calculate how many moles of water and isopropyl alcohol there are in the mixture.
Equation 3:m = mass of substance [g]
n = amount of substance measured in moles [mol]
Using Equation 3, the amount of water (in moles) is:
n = m/M = 9.98 g / 18 g/mol = 0.55 molSimilarly, the amount of isopropyl alcohol (in moles) is:
n = m/M = 39.25 g / 60 g/mol = 0.65 molThe total moles in the mixture are:
ntotal = nH2O + nisopropyl alcohol = 0.55 mol + 0.65 mol = 1.2 molWe know from Equation 1 in the Introduction that the mole fraction is defined as the ratio of the number of moles of a particular substance to the total number of moles of all the substances in a mixture.
Equation 1:where
= mole fraction of the substance "i"= number of moles of the substance "i"
= total number of moles of all substances in the mixture
Using Equation 1, we can calculate the mole fraction of isopropyl alcohol in the mixture:
xisopropyl alcohol = nisopropyl alcohol / ntotal = 0.65 mol/1.2 mol = 0.54You can use this example calculation to calculate the mole fraction of isopropyl alcohol for all the mixing ratios you are testing in your experiment.
Ask an Expert
Global Goals
The United Nations Sustainable Development Goals (UNSDGs) are a blueprint to achieve a better and more sustainable future for all.
Variations
- In this project, you measured the mass and the volume of each of your solutions. One other parameter you could measure is the temperature. You want to start out with the same temperature for both liquids. But once you mix the water and the isopropyl alcohol, does the temperature of the mixture change? Find out by yourself!
- Explore the mixing behavior of other miscible liquids. Do their mixtures result in an excess volume?
- Expand the project by trying other mixing ratios of water and isopropyl alcohol that were not listed in Table 1.
- The excess volume is not only dependent on the mixing ratio of the two liquids but also on their temperatures. Repeat the experiment with just one mixing ratio, but test a range of different temperatures for the two liquids. How does the excess volume change with temperature?
Careers
If you like this project, you might enjoy exploring these related careers: