Abstract
Does your hair go crazy when the weather turns damp? Did you know that strands of hair can relax and lengthen when the humidity increases and then contract again when the humidity decreases? In fact, hair strands can be used as the basis for a hygrometer, a device which measures the humidity level in the air. Will a hygrometer help you to predict bad hair days(!) or can you use it to help predict the weather?Summary
Sources
- The information on how to build a hygrometer is from:
The Franklin Institute. (n.d.). Make Your Own Hygrometer. Retrieved February 19, 2015.

Objective
To make and use a hygrometer, an instrument for measuring the level of humidity in the air.Introduction
Changes in humidity are an important part of daily weather. In this science fair project you will build a hydrometer, an instrument used to measure the humidity level in the air. Your hygrometer will use strands of human hair as its sensing element.
In order to understand how a hygrometer made with strands of hair works, it is important to understand the structure of a human hair shaft. (The hair shaft is the part of the hair strand that sticks out above the surface of the skin, while the hair follicle is the specialized structure that is in the skin that produces each individual hair strand.) Figure 1, illustrates the microscopic structure of a human hair shaft. The left-hand panel of the illustration (Figure 1a) is a cartoon of a human hair shaft with a cut-away view to show the inner structure. Each strand of hair has several outer layers of flattened cuticle cells (Cu), which surround the fibrous cortical cells (Co) (they make up the cortex). The medulla (Md) is a central core of cells in the hair shaft. Also shown is a microfibril (MF) within a cortical cell. The middle panel (Figure 1b), shows an actual hair shaft under the microscope. You can see how the flattened cuticle cells (Cu) have a scale-like appearance when magnified. The dark central medulla (Md) is also visible. The right-hand panel (Figure 1c), shows a cross-section of a fine human hair. Here you can see that the cuticle cells (Cu) are highly flattened, and wrap around the cortical cells (Co) in many layers. The cortical cells contain the dark pigment granules that give each hair strand its natural color.
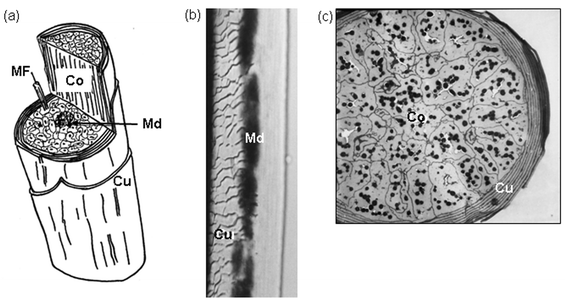
The inner structure of a strand of human hair is comprised of a small core called a medula that is surrounded by thick walls of cortical cells and microfibril. The entire hair shaft is also covered by a thin layer of cuticle cells.
Figure 1. Microscopic structure of a human hair shaft. Part (a) shows a cutaway cartoon of a single hair shaft. The labels show cuticle cells (Cu), cortical cells (Co), the medulla (Md), and a microfibril (MF) within a cortical cell. Part (b) shows a transmitted light micrograph of a single hair strand. The scale-like layer of cuticle cells (Cu) is clearly visible, as is the central medulla (Md). Part (c) shows a cross-section of a fine hair strand. The flattened cuticle cells (Cu) wrap tightly around the cortical cells (Co), which contain many dark pigment granules (Tobin, 2006).
The cuticle cells have a coating of specialized molecules that repel water. These molecules are called lipids. By repelling water, the lipid molecules help to protect the hair strand.
The predominant proteins in hair are from the family of keratins, the same family of proteins that make your fingernails. Protein molecules are built from amino acids. In a hair strand, the keratin molecules are held together by two main forces: chemical cross-links between cysteines (a type of amino acid that is common in these keratin molecules) and weaker hydrogen bonds. The keratins in hair have many such bonds, making a hair strand strong and flexible. Each cysteine in the keratin molecule is a potential attachment point, where the keratin molecule can be tightly connected to another cysteine, forming a chemical bond called a cross-link. The many weaker hydrogen bonds between the keratin molecules can be disrupted by water, including water vapor in the air, with each keratin binding to water instead of each other. In fact, this is what causes hair strands to expand in moist air and to contract in dry air.
"A single lock will stretch about two and a half percent as the humidity goes from 0 to 100 percent. While that is only a smidgen of length, the rate of this change is very dependable, so it is possible to obtain highly accurate humidity readings by measuring these tiny shifts. Hair hygrometers are best suited for keeping track of humidity in closed settings such as office buildings, where the variations are small and the goal is to keep humidity constant." (Weather Notebook, 2005)
In this science project, you will build a hair hygrometer and use it to measure humidity changes inside your home, or outside to help you predict the weather.
Terms and Concepts
- Humidity
- Hygrometer
- Hair shaft
- Hair follicle
- Cuticle cells
- Cortical cells or cortex
- Medulla
- Lipids
- Keratins
- Cross-links
- Hydrogen bonds
Questions
- How does the structure of the hair strand change as the relative humidity of the surrounding air varies?
- How humid do you think it is in the bathroom after a shower? What about in another room in your home?
Bibliography
For information on hygrometers and relative humidity, see these references:- The Franklin Institute. (n.d.). Make Your Own Hygrometer. Retrieved February 19, 2015.
- American Meteorological Society. (n.d.). Hair Hygrometer. Glossary of Meteorology. Retrieved March 27, 2014.
- Swiss Institute of Bioinformatics. (2006, May 22). Hair keratin. Protein Spotlight. Retrieved March 16, 2012.
- The Science Company. (n.d.). Understanding Relative Humidity and the Hygrometer. Retrieved March 16, 2012.
- Tobin, D.J. (2006, October 26). Biochemistry of Human Skin -Our Brain on the Outside. Chemical Society Reviews. Retrieved July 10, 2020.
Materials and Equipment
- 70% isopropyl rubbing alcohol
- Measuring spoons
- A small bowl
- Long strands of human hair, about 20 cm long each (9)
- Cotton swabs
- Metric measuring tape or ruler
- Flat pieces of plastic, about 8 cm x 8 cm and thin enough that you can easily cut them, such as from the lid of a deli container (3)
- Scissors
- Dimes (3)
- Tape
- Small nails (6)
- Elmer's school glue
- Scrap pieces of wood or flat polystyrene, about 25 cm x 10 cm (3)
- A hammer
- Pencil or pen
- Hair dryer
- A plastic box that can seal and that is large enough to fit the wood or Styrofoam piece inside of it
- Wet sponge, paper towel, or small towel
- Optional: Psychrometer or electric hygrometer.
- You can purchase a psychrometer from online suppliers such as Carolina Biological Supply Company or you can make one using the Science Buddies science project idea Make Your Own Psychrometer.
- Lab notebook
Disclaimer: Science Buddies participates in affiliate programs with Home Science Tools®, Amazon.com, Carolina Biological, and Jameco Electronics. Proceeds from the affiliate programs help support Science Buddies, a 501(c)(3) public charity, and keep our resources free for everyone. Our top priority is student learning. If you have any comments (positive or negative) related to purchases you've made for science projects from recommendations on our site, please let us know. Write to us at [email protected].
Experimental Procedure

- Figure 2 shows what the completed hygrometer looks like. Look at the figure to get an idea of how you will be making your hygrometers.
- For this science project you will be making three hair hygrometers, all following the same pattern. Testing multiple hygrometers will help you ensure that your results are accurate and reproducible.
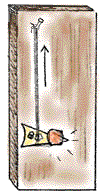
Figure 2. A simple hygrometer made with strands of human hair. The hair cells expand when the humidity is higher, and the weight of the dime moves the pointer lower. The hair cells contract when the humidity is lower, and the contracting hair pulls the pointer higher. (Image courtesy of The Franklin Institute Resources for Science Learning).
- Wipe down the strands of hair with diluted isopropyl rubbing alcohol.
- Make a solution of 25% rubbing alcohol and 75% water. You can do this by mixing one tablespoon of water with one teaspoon of rubbing alcohol in a small bowl.
- Use a dab of this 25% rubbing alcohol solution on a cotton swab to wipe down the hair strands.
- This will remove any residual oils and allow water to permeate the hair strands more easily.
- Set the hair strands aside somewhere where you will not easily lose them.
- Use the template shown in Figure 3, to cut each piece of plastic into a triangular shape to make the pointer.
- Make three pointers total and make sure each of your pointers is the exact same size.
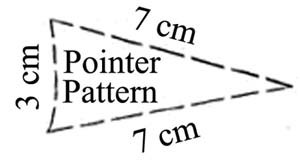
Figure 3. Template for making the pointer, with lengths shown in centimeters (cm). (Image courtesy of The Franklin Institute Resources for Science Learning, www.fi.edu/learn.)
- Tape a dime onto each pointer, as shown in Figure 4, centered about 4.5 cm from the left side of the pointer.
- Some of the dime will stick out a little above and a little below the two long sides of the pointer.
- Again, make sure that you tape the dime in the exact same place on each pointer.
- What do you think the purpose of the dime is?
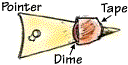
Figure 4. A close-up of the pointer, with dime attached. (Image courtesy of The Franklin Institute Resources for Science Learning, www.fi.edu/learn.)
- Poke one of the nails through one of the pointers, about ½ cm from the left side of the pointer. Wiggle the nail until the pointer moves freely and loosely around the nail. Do this to each pointer. Again, make sure to use the exact same position on each pointer.
- Why do you think it is important that the pointer can move freely around the nail?
- To each pointer glue three hair strands, between the dime and the nail hole, about 1 cm to 1.5 cm to the right of the nail hole. As usual, use the same position for each of the pointers.
- Position each pointer on one of the wood or polystyrene bases about three quarters of the way down the side (about 19 cm down) with the left side of the pointer about 1 cm from the left side of the base. Carefully hammer a nail through each pointer's nail hole and securely into the base. As usual, use the same position for each of the hygrometers.
- Make sure the pointer can still turn easily around the nail.
- Carefully hammer in another nail about 2.5 cm from the top of each base, in line with where the hair was glued to the pointer. As usual, use the same position for each of the hygrometers.
- Gently pull the hair strands taut, so that the pointer points parallel to the ground. That is, make sure the point of the pointer is perpendicular to the hair. The hair should hang perfectly vertical and the pointer should point perfectly horizontal.
- Glue the free ends of the hair strands to the top nail on each base. You may need to hold the hair strands taut while the glue starts to dry so that they dry in a taut position.
- If the hair is too long, trim the ends.
- Make sure that the glue has completely dried before going on to the next step.
- You should now have three hair hygrometers. You will want to calibrate each one using the two extremes on your hygrometer scale, 0% and 100% humidity.
- For 0% humidity, dry the strands gently with a hair dryer until the strands no longer decrease in length (the pointer no longer moves). Mark this location as 0%.
- For 100% humidity, close up the hygrometer in a plastic box with a wet sponge or paper towel. Wait at least ten minutes. Mark the pointer location lightly, re-seal the box, and wait another ten minutes. Keep doing this until the pointer location is no longer changing. Mark the final pointer location as 100%.
- For intermediate values, you could just divide the space between 0% and 100% into five equal increments and mark the locations for 20, 40, 60, and 80%, then divide each of these in two to mark locations for 10, 30, 50, 70, and 90%.
- Because the hair strands may have changed only a little bit in length between the 0% and 100% humidity points, you may not have much room to write in 10, 30, 50, 70, and 90% marks. If this is the case, you can estimate these marks when doing your measurements based on the positions of the other marks.
- Although calibrating the hair hygrometers this way may not make them accurate to 10%, this calibration can still give you a rough approximation of percent humidity. The best way to calibrate your hair hygrometer would be to compare it to another instrument that measures humidity, specifically a psychrometer or electric hygrometer.
- You can purchase a psychrometer from online suppliers such as Carolina Biological Supply Company or you can make one using the Science Buddies science project idea Make Your Own Psychrometer.
- Test how your hair hygrometers respond to changes in humidity and how they can be used to measure different humidity levels. Make a data table in your lab notebook to record the humidity levels you measure for each hygrometer in different locations or conditions.
- Put your hygrometers in a room in your home for 10 minutes. How humid do they say it is in the room? What factors do you think might affect how humid the room is? Write your observations in your lab notebook.
- Put your hygrometers in a bathroom while the shower is running. How humid do they say the bathroom is after the shower has run for several minutes?
- Put your hygrometers outdoors in an area that is sheltered from precipitation. Monitor the hygrometers regularly throughout the day (such as once every hour) over the course of several days, writing your observations in your lab notebook. How does the humidity change during the course of a "normal" day? You can make a graph of your results by putting the time on the x-axis and the measured percent humidity on the y-axis. How does the humidity change when a storm system moves through your area? How do the humidity levels outside compare to the humidity levels you measured inside?
- For all of the humidity readings you take, do all three hygrometers give similar readings, or are they very different? If there are differences, what do you think might cause them?
Ask an Expert
Variations
- For an experiment that compares hygrometers made with untreated and chemically lightened hair, see the Science Buddies project Does Chemical Lightening Affect the Structure of Human Hair?
- Using the hair hygrometers you made for this project, you could compare how the humidity inside your home compares to the humidity outside over the course of a day. Put one hygrometer outdoors in an area that is sheltered from precipitation and put one hygrometer inside your home (in a room other than the bathroom). Monitor the hygrometers throughout the day and record your observations. Over the course of the day, do the humidity levels in the house change in a way that is similar to how they change outdoors? In what ways are they different? Record your observations over multiple days to see if the trends stay the same or if they change.
- For a different method of measuring humidity, see the Science Buddies project Make Your Own Psychrometer. How do the two methods compare?
Careers
If you like this project, you might enjoy exploring these related careers:








