Others Like “Swimming in Acid: Understanding Ocean Acidification” (top 20 results)
|
The rates of some chemical reactions can actually be increased by adding light. Light sometimes interacts with one or more of the chemicals and provides an "energy boost" that dramatically speeds up a normally slow reaction. In this photochemistry science project, you will experiment with the effect of light on a chemical reaction. The reaction converts iodine, which forms a dark-orange solution, to iodide, which is colorless!
Read more
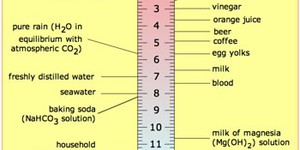
This experiment is for all the kids out there who love boiled cabbage! You say you do not like cabbage? Well maybe you will like this amazing color-changing liquid you can make with cabbage. Which solutions around your house can make the cabbage juice change color? Find out while you learn about acids and bases and how to test for them.
Read more
There is nothing quite like the smell of fresh-baked bread to make your mouth water! As any baker can tell you, you cannot bake bread without yeast. Yeast actually eat sugar so that they can reproduce and make more yeast, and make bread dough rise. But can they use sugar substitutes to do this? In this science project you will get to investigate how well yeast grow with sugar substitutes as a food source. Pass the butter, please!
Read more
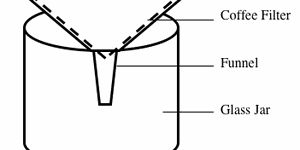
Have you ever mixed together salt and sand? It is fun to see how all of those tiny grains of salt and sand mix together! But what if you had to separate them out again? Do you have nightmares of tiny tweezers, a magnifying glass, and hours spent picking grains of salt and sand apart? Do not be afraid, there is another way! In this chemistry science project you will use the differences in solubility between salt and sand to find out the simple "solution" to this problem.
Read more
Alka-Seltzer® tablets fizzle furiously when dropped into water. The moment the tablet starts dissolving, a chemical reaction occurs that releases carbon dioxide gas. In this science project, you can even measure how long and loudly your tablet fizzes using a smartphone equipped with a sensor app. Do you think you can make Alka-Seltzer fizz faster or more loudly by changing the temperature of the water? How big of a difference in the rate of a chemical reaction can temperature make?
Read more
You have probably heard the saying that "water and electricity don't mix." Well, in this chemistry science fair project you will mix them, to create two solutions, one basic and one acidic. The apparatus is very simple, but the chemistry is complex and offers many avenues for exploration.
Read more
Do you want your hair to be shiny after you wash it? Do you want your clothes to stay bright and soft after laundering, and last a long time? You might think that a special shampoo or detergent can make this happen, but in this chemistry science fair project, you'll discover that how well people and things get clean has a lot more to do with the type of water used for washing than any special shampoo or soap. Did you know that water can be classified as either soft or hard? Soft water lathers…
Read more
Have you ever wondered how antibiotics and other medicines are able to stop dangerous infections? How do such medications kill microorganisms without in general harming the person the microorganisms are infecting? Because many different types of microorganisms can infect us, we have had to develop an amazing number of ways to deal with these harmful microbes. Fungal infections can be particularly dangerous, but we have developed many different antifungal medications that can usually deal with…
Read more
Have you ever had a refreshing bath using a bath bomb? A bath bomb is several ingredients mixed and molded into a shape, which becomes fizzy when it touches the water. It can be quite a relaxing experience, especially if your bath bomb has a nice fragrance or includes some bath salts. The fizz is the result of a chemical reaction taking place between different ingredients within the bath bomb. In this science project, you will get to make your own homemade bath bombs and explore how changing…
Read more
Have you ever left your bike outside in the rain? If so, you might have discovered unpleasant surprises afterwards—reddish-brown patches, known as rust, and your wheels, brakes, and gears might have stopped working so smoothly. In this chemistry science fair project, you'll learn why rust, a type of corrosion, is a serious problem. You'll also discover that not all rains are the same! Find out which ones can speed up the rusting process.
Read more
|
Explore Our Science Videos
How to Build an ArtBot
Mold Hard Boiled Eggs
Build A River Model – STEM Activity