Creating a Kidney: How Stem Cells Might Be Used to Bioengineer a Vital Organ
Abstract
The human kidney is the most commonly transplanted organ in the United States, numbering more than 17,000 transplants in 2010 alone! But kidney transplantation technology faces a lot of challenges, including a shortage of kidney donors and the need for recipients to take immunosuppressant drugs to keep their bodies from rejecting a transplanted kidney. In this science project, with the help of bioinformatics databases, you will explore how a kidney could be bioengineered using stem cells, potentially overcoming these problems.Summary
Teisha Rowland, PhD, Science Buddies

Objective
Use bioinformatics databases to determine the best protein environment for bioengineering a kidney.
Introduction
The demand for kidney transplants is high, but human donors are limited. And even when transplants are successful, to keep the body from rejecting the transplanted organ, the recipient must take long-term immunosuppressant drugs that can cause a number of undesirable side-effects. Ideally, a recipient should receive a patient-specific kidney, made from the recipient's own cells, which would therefore require neither a donor nor immunosuppressant drugs. But how could such a patient-specific kidney be made?
Bioengineering has had some success at creating and transplanting organs into patients. Bioengineering focuses on how tissues and organs can be made, or engineered, to replace damaged or lost tissues and organs. It is just one part of the larger field of regenerative medicine, a fast-growing field focused on creating ways to replace or heal damaged tissues or organs. In 2008, bioengineers achieved the first successful transplant using an organ engineered with a patient's own stem cells, which are cells that can turn into a number of different cell types and can proliferate (multiply in numbers). Specifically, a patient-specific bronchus (an airway tube that goes to a lung) was bioengineered using the patient's stem cells and a trachea from a cadaver (deceased donor). Some other organs (such as urethras) and tissues have been bioengineered using a patient's cells and a synthetic scaffold. A synthetic scaffold is a structure that can be synthesized and supports the growth of cells. If a synthetic scaffold is used instead of cadaver tissue, it makes donors unnecessary. Watch this video to learn more about some efforts to bioengineer tissues and organs.
Overall, efforts to bioengineer entire organs have been limited; bioengineering an organ such as the kidney would be a huge undertaking. The kidney is a complex organ, with approximately 18 different cell types. Instead of bioengineering the entire organ, a more feasible goal may be to bioengineer the kidney's nephrons, the main functional units of the kidney. Every kidney contains approximately 0.5 million to 1 million nephrons, and they filter waste from the blood, expel that waste in the form of urine, and return clean fluid to the bloodstream. Every nephron is made up of two main parts, the renal corpuscle and the renal tubule, as shown in Figure 1. The renal corpuscle receives unfiltered blood through the glomerulus and filters it through the Bowman's capsule, which sends the waste liquid to the renal tubule, from which it is sent on to be expelled eventually as urine.
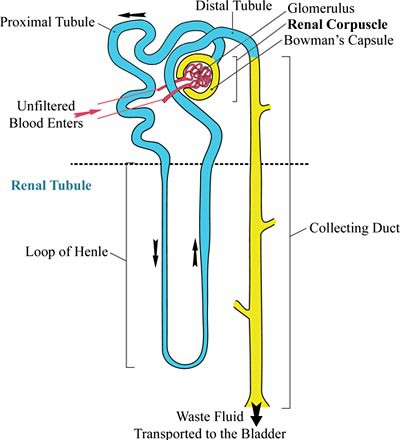 Image Credit: Wikimedia Commons / GNU Free Documentation License
Image Credit: Wikimedia Commons / GNU Free Documentation LicenseDiagram of a kidney. In a kidney nephron unfiltered blood enters through the Bowman's capsule and glomerulus. From there, it moves along the proximal tubule and down into the renal tubule where it is pumped back up to the distal tubule. The final area is a collecting duct where waste fluid is eventually transported into the bladder.
Figure 1. A nephron is made up of two main parts: the renal corpuscle and the renal tubule. In the renal corpuscle, the glomerulus receives unfiltered blood, which is filtered through the Bowman's capsule. From there, the blood goes through the renal tubule (colored blue in this diagram), which consists of the proximal tubule, loop of Henle, and distal tubule. The waste liquid is then transported to the collecting duct, from which it goes to the bladder and is expelled as urine. (Adapted from Yosi I (2007, December 25), Wikimedia.)
While commonly used to bioengineer organs, stem cells come in many varieties and some work better in certain situations than others. Some stem cells have been found in the adult kidney, but they are limited; they are thought to be dormant unless a minor repair is needed, and if the nephrons in an adult human kidney are damaged or lost, it is thought they cannot be regenerated. However, embryonic stem cells (or ESCs) and induced pluripotent stem cells (or iPSCs) have been found able to transform, or differentiate, into different kidney cell types. Human embryonic stem cells (hESCs) are isolated from early-stage embryos that have only about 150 cells. (To read more about hESCs, see the related Project Idea, Taking Short Cuts: How Direct Reprogramming Can Transform One Type of Cell Straight into Another.) Because hESCs are isolated at such an early stage during development, they can multiply and become any cell type in the human body, whether bone, hair, heart, etc. Cells such as hESCs that can give rise to any cell type in the body are referred to as pluripotent (from the Latin words plurimus, meaning "very many," and potens, meaning "having power").
Human induced pluripotent stem cells (hiPSCs) are cells that were mature, adult human cells but that have been "reprogrammed" to behave as hESCs. Consequently, not only are hiPSCs pluripotent, like hESCs, but hiPSCs can also be patient specific. This means that a patient could have some of his or her cells removed, such as skin or blood cells, reprogrammed into hiPSCs, and then have those cells turned into whatever types the patient needs, such as kidney cells for bioengineering a new kidney. Making patient-specific cells like this may avoid some potential complications that can occur with tissue and organ transplants, such as immune rejection, where the recipient's immune system tries to prevent the transplanted organ or tissue from surviving.Although iPSCs have proved capable of differentiating into different kidney cell types, current techniques are inefficient and have not been thoroughly tested with hiPSCs (they have been used primarily with mouse iPSCs). Specifically, the techniques usually lead to a mixture of cells, not a purified population of a certain kidney cell type, and the techniques are generally slow. How can these techniques be improved? One way is by intelligently altering the cells' growing conditions, also called the cells' microenvironment. In a laboratory, the hiPSCs' microenvironment is defined by two key factors: the liquid the cells are grown in, called the cell media, and the substrate the cells are grown on, which can include other cells called "feeder" cells. Altering these two factors can affect how well the hiPSCs differentiate and what cell type they differentiate into. Nutrients and growth factors (substances the cells need to grow) are supplied to the hiPSCs from their cell media, while the cells grab onto the substrate to stay in place. Watch this video to learn more about how hiPSCs are cultured.
To differentiate hiPSCs into a certain cell type, researchers often try to manipulate the cells' microenvironment so that it mimics what the cell would normally be exposed to in the human body. This means that if you want to make kidney cells, the hiPSC media may include soluble growth factors (or "soluble factors") that the cells might encounter during normal kidney development. Likewise, the substrate on which the hiPSCs are differentiating may be made to mimic the structural part of the microenvironment that the kidney cells would encounter in the human body. The extracellular matrix (ECM) is a mesh-like network of proteins that surrounds cells in the body, giving structural support to the cells and promoting communication among them. Some of the proteins that make up the ECM include laminins, collagens, vitronectin, and fibronectin. To interact with any one of these proteins, the hiPSCs need specific cell receptors on the surface of their cells. Consequently, by looking at which cell receptors certain cells have, such as developing kidney cells, researchers can obtain data on what kinds of ECM proteins these cells need as a substrate.
What microenvironment will be the best one for turning hiPSCs into kidney cells? How can this knowledge be applied to bioengineer patient-specific nephrons, and eventually kidneys? In this bioengineering science project, you will be using online bioinformatics databases (collections of biological data) to explore how researchers select soluble factors and suitable ECM proteins to guide hiPSCs into differentiating into nephron cells, and how this could lead ultimately to bioengineering a kidney.
Terms and Concepts
- Patient-specific
- Bioengineering
- Regenerative medicine
- Stem cells
- Synthetic scaffold
- Nephrons
- Renal corpuscle
- Glomerulus
- Bowman's capsule
- Renal tubule
- Differentiate
- Human embryonic stem cells
- Pluripotent
- Human induced pluripotent stem cells
- Immune rejection
- Microenvironment
- Cell media
- Substrate
- Growth factors
- Soluble factors
- Extracellular matrix (ECM)
- Cell receptors
- Bioinformatics
- In vivo
- In vitro
- Signaling pathways
- Apoptosis
- Epithelial-to-mesenchymal transition (EMT)
- Precursor cells
- Integrins
- Microarray
Questions
- What are the two main parts of a kidney's nephron? What are their functions?
- Why is it ideal to bioengineer organs that are patient-specific?
- What makes stem cells so useful in bioengineering organs?
- What is the field of bioinformatics? How can it help researchers find out information without doing experiments in a laboratory setting?
Bibliography
To perform this bioengineering science project, you will need to use some of these databases:
- Memorial Sloan-Kettering Cancer Center. (n.d.). Pathway Commons. University of Toronto. Retrieved November 6, 2014.
- R&D Systems. (n.d.). Embryonic and Induced Pluripotent Stem Cells & Lineage-specific Markers. Retrieved November 6, 2014.
- National Center for Biotechnology Information (NCBI). (n.d.). Gene. Retrieved June 28, 2012.
These resources are good places to start gathering information about stem cells and bioengineering organs:
- National Institutes of Health (NIH). (2009, April 28). Stem cell information: Stem cell basics. Retrieved June 28, 2012.
- The New York Stem Cell Foundation. (n.d.). Stem Cell Glossary. Retrieved June 20, 2023.
The 2008 study on the first successful organ transplant bioengineered with a patient's own stem cells:
- Macchiarini, P., et al. (n.d.). Clinical transplantation of a tissue-engineered airway. Retrieved April 30, 2018.
Information about the soluble factors in Table 1 was taken from these sources (also listed in Table 1):
- Kramer, J., et al. (2005, December 19). Cells differentiated from mouse embryonic stem cells via embryoid bodies express renal marker molecules. Retrieved June 28, 2012.
- Ren, X., et al. (2010, May 6). Differentiation of murine embryonic stem cells toward renal lineages by conditioned medium from ureteric bud cells in vitro. Retrieved June 28, 2012.
- Kobayashi, T., et al. (2005, August 26). Wnt4-transformed mouse embryonic stem cells differentiate into renal tubular cells. Retrieved June 28, 2012.
- Morizane, R., et al. (2009, October 31). Differentiation of murine embryonic stem and induced pluripotent stem cells to renal lineage in vitro. Retrieved June 28, 2012.
- Dankers, P., et al. (2011, February 18). From kidney development to drug delivery and tissue engineering strategies in renal regenerative medicine. Retrieved June 28, 2012.
Materials and Equipment
- Computer with an Internet connection
- Lab notebook
Experimental Procedure
Driving Kidney Cell Differentiation Using Soluble Factors
What soluble factors would be best to use when trying to differentiate human induced pluripotent stem cells (hiPSCs) into kidney cells, specifically the kidney cells of the nephron, the functional unit of the kidney? To differentiate hiPSCs into nephron cells efficiently, the soluble factors that are added to the cell media need to prompt the hiPSCs to turn into nephron cells. A few studies have investigated differentiating iPSCs into nephron cell types, but the techniques are inefficient, resulting in a mixture of cell types with only 60% to 90% being the desired cell type. In this part of the science project, you will decide which soluble factors, and combinations of soluble factors, would be best to test when prompting hiPSCs to turn into different types of nephron cells.
- Look at Table 1, which summarizes the soluble factors used in key publications of research on turning ESCs or iPSCs into different nephron cell types. Note: Table 1 summarizes this information so you do not need the actual papers, which are not publicly available, to do this bioengineering science project.
- What soluble factors did the different researchers add to make either podocytes (cells in the glomerulus) or renal tubule cells? You can look at Figure 1 (in the Introduction in the Background tab) to review the structure of, and different cell types in, the nephron.
- For example, two groups of researchers (Kramer et al., 2005, and Ren et al., 2010) were able to make podocyte cells. One of these groups (Kramer et al., 2005) did not report adding any soluble factors, while the other one (Ren et al., 2010) reported adding two different soluble factors, activin A and retinoic acid (RA). Three groups (Kramer et al., 2005; Kobayashi et al., 2005; and Morizane et al., 2009) made renal tubule cells using one, two, or no soluble factors.
- Although one research group (Kramer et al., 2005) added no soluble factors, whether their efficiency could be improved by adding soluble factors should be investigated.
- In the bottom row of Table 1, where the Dankers et al. research from 2011 is listed, several soluble factors are named that are necessary for embryonic kidney development in the body, also called in vivo. These factors are not known to have been tested on cells grown in a laboratory, or in vitro, but they are likely candidates for prompting hiPSCs to differentiate into some nephron-like cells.
- Note: Extensive studies have not yet been published on turning pluripotent stem cells into cells of the Bowman's capsule, and so it is omitted from this bioengineering science project. See the "Variations" for how you could explore bioengineering this part of the nephron.
- Although you do not need the research papers to do this science project, if you want to learn more about a particular paper, click on the link under the "Research Paper" column to see the paper, as an abstract or in its entirety. What can you understand about the paper and its reported findings by reading the abstract?
- If you are interested in ways to obtain these papers, you may be able to use the guide Resources for Finding and Accessing Scientific Papers to do so.
- If you choose to read the papers, you may also find you need some help from someone experienced with stem cells or bioengineering to help you find out How to Read a Scientific Paper.
- What soluble factors did the different researchers add to make either podocytes (cells in the glomerulus) or renal tubule cells? You can look at Figure 1 (in the Introduction in the Background tab) to review the structure of, and different cell types in, the nephron.
| Nephron Cell Type | Main Supplemented Soluble Factors | Research Paper |
| Renal Corpuscle Cells: Podocytes in the Glomerulus | No factors added | Kramer et al., 2005 |
|
Ren et al., 2010 | |
| Renal Tubule Cells | No factors added | Kramer et al., 2005 |
|
Kobayashi et al., 2005 | |
| Activin A | Morizane et al., 2009 | |
| Necessary for General Kidney Development |
|
Dankers et al., 2011 |
*Wnt4 was not actually a soluble factor, but was transfected into the cells so that the cells made their own Wnt4. In this bioengineering science project, Wnt4 can still be explored and treated as a soluble factor.
** Conditioned media is made by exposing media to cells so that factors secreted by the cells are collected in the media. In the Ren et al. paper from 2010, conditioned media was made and tested from ureteric buds, which are the structures that develop into the entire kidney, except for the nephrons. For the purposes of this engineering science project, conditioned media can be ignored, but it can be explored in the Variations section.
- Learn more about the soluble factors in Table 1 by using at least two of the following databases (listed in steps 2.a.–2.c.). Before you start using the databases, make a data table in your lab notebook like Table 2, below, for you to organize the information that you will be collecting on the different soluble factors. What are the functions of these different soluble factors? How might they be used to differentiate stem cells into different kidney cell types? Note that you may be unable to answer all of the questions in Table 2 for all of the soluble factors, and that not every database may have information on every soluble factor, but put as much relevant information into your data table as you can as you search through the databases.
- The NCBI Gene Database:
- Use the Science Buddies NCBI Gene & SNP Tutorial to help you navigate the NCBI Gene Database.
- For your searches, use the abbreviated gene names listed in Table 2, below.
- Make sure to choose the top search result that lists your soluble factor name, such as "INHBA," and that it is for humans (Homo sapiens).
- What does the database tell you about each soluble factor? Fill in your data table with any relevant information from the NCBI Gene database.
- For example, depending on the information provided for a given soluble factor, you might fill in the data table as follows: Under "Does it regulate proliferation or apoptosis?" you may write "Yes, inhibits cell growth and proliferation." Under "Does it regulate differentiation?" you may write "Yes, regulates cell differentiation." Under "What other key functions does it have?" you may write "Regulates stem cell maintenance and progesterone production."
- If you run across terms you are unfamiliar with (such as possibly "progesterone"), you should always look them up and try to figure out how they fit in with what else you know about the soluble factor.
- Pathway Commons:
- In the search bar on the homepage, type in the abbreviated gene name of the soluble factor you want to explore (such as "INHBA").
- On the results page, choose the "interactions" option. This will take you to a network visualizer which visually shows the connections between the soluble factor and other genes/factors. The soluble factor you searched for will be in the middle of the interaction graph.
- Click on the circle with the soluble factor's name. This should cause text to pop with summary information about the soluble factor.
- You can repeat this process to learn more about the other factors shown in the network visualizer to interact with the soluble factor you searched for.
- What other factors does the factor you searched for directly regulate, and which ones does it more distantly regulate?
- Can you tell how they regulate each other?
- Fill in your data table with any relevant information from the Pathway Commons on the different soluble factors.
- Embryonic and Induced Pluripotent Stem Cells & Lineage-specific Markers:
- This gives a visual overview of lineage-specific markers during the differentiation of pluripotent stem cells. Click on any factor's name (and then click on its name in the window that pops up) to be taken to a webpage with more information on it.
- Fill in your data table with any relevant information from this database on the different soluble factors.
- The NCBI Gene Database:
| Soluble factor | Abbreviated gene name | Family it belongs to | Does it regulate proliferation or apoptosis?* | Does it regulate differentiation? | Does it regulate development? | Does it regulate epithelial-to-mesenchymal transition?** | What other key functions does it have? |
| Activin A | INHBA | ||||||
| Retinoic acid | RARA | ||||||
| Wnt4 | WNT4 | ||||||
| Hepatocyte growth factor | HGF | ||||||
| Transforming growth factor alpha | TGFA | ||||||
| Transforming growth factor beta | TGFB1 | ||||||
| Insulin-like growth factor | IGF1 | ||||||
| Platelet-derived growth factor | PDGFA |
*Apoptosis is programmed cell death.
** During development, the tubules in the nephron (and many other tissues throughout the body) undergo a process called epithelial-to-mesenchymal transition, or EMT, where one type of tissue (epithelial tissues) differentiates into another type (mesenchymal tissues).
- After you have filled out your data table of soluble factors using at least two of the databases in step 2, look at how different and how similar the soluble factors are.
- Do many of the soluble factors regulate the same function, such as differentiation? Do any factors regulate three of the same functions? If any two factors regulate three of the same functions, they may act similarly.
- Review Table 1 to see which soluble factors have been used previously in the named studies in certain combinations. Can you substitute any of the factors from a previously used combination with a similar factor, based on your data table?
- For example, Wnt4, HGF, and Activin A have been previously used in a combination (Kobayashi et al., 2005). Is there another factor similar to Wnt4 that you could use instead of Wnt4 in this combination? Or is there another factor similar to HGF or Activin A that could be substituted instead?
- In your lab notebook, create a data table of the different combinations that could be created based on how similar the soluble factors are.
- Make columns listing the "Nephron Cell Type" and "Existing Combinations of Soluble Factors," as listed in Table 1. Then include columns listing the "Substitutions" that could be made for each factor and the "Potential Combinations" that could be made based on the substitutions (as discussed in step 5a for Wnt4, HGF, and Activin A).
- Using different combinations of soluble factors may improve upon the current nephron cell differentiation protocols discussed in Table 1. Based on the data table you made in step 2, what would be feasible, new soluble factor combinations to test to turn hiPSCs into podocytes or renal tubule cells? Do you expect some combinations to work better than others? Why? Might some combinations work well to make nephron cells, or nephron precursors (cells that can easily turn into nephron cells), in general?
Using the Extracellular Matrix to Drive Kidney Cell Differentiation
What Extracellular Matrix (ECM) proteins would be best to use when trying to differentiate hiPSCs into nephron cells? The ECM is a mesh-like network of proteins that surrounds cells in the body, giving structural support to the cells and promoting communication among them. To efficiently differentiate hiPSCs into nephron cells, not only do the hiPSCs need the appropriate soluble factors in their media, but the substrate they are grown on can also affect how they differentiate. (The studies summarized in Table 1 did not investigate different substrates, but generated nephron cell types by initially creating cell formations called embryoid bodies, which were cultured by floating them in the media, without touching a substrate, for two to five days. After being in the floating culture, they were cultured on gelatin, which is a standard culture substrate made up of fragments of collagen.) Differentiating hiPSCs on certain ECM proteins may produce nephron cells more efficiently, and this research could be used as the basis for designing synthetic scaffolds on which to grow kidney cells for transplants. To interact with ECM proteins (including laminins, collagens, vitronectin, fibronectin, and more), the hiPSCs need specific cell receptors on the surface of their cells. In this part of the science project, you will examine which cell receptors, called integrins, are made by kidney cells during development.
- Table 3 lists integrins and the ECM proteins they bind.
- Integrins are heterodimers, meaning they are made up of two different subunits, specifically an alpha and a beta subunit. Only certain alpha and beta subunits can combine. The combination determines what ECM proteins they can bind.
- Note: Not all integrin subunits are listed in Table 3. Table 3 includes integrin subunits that have data in the NIH Gene database and that form integrin heterodimers with well-established information on the ECM proteins they bind. Also, the integrin heterodimers in Table 3 may bind additional ECM proteins that are not listed.
| α Integrin Subunits | β Integrin Subunits | ECM Proteins They Bind |
| α2 | β1 | Laminin-111, Laminin-211, Collagen I, Collagen IV |
| α3 | β1 | Laminin-332, Laminin-511, Laminin-521, Collagen IV, Fibronectin |
| α5 | β1 | Fibronectin, Fibrinogen |
| α6 | β1 | Laminin-111, Laminin-211, Laminin-221, Laminin-332, Laminin-511, Laminin-521 |
| β4 | Laminins-332, Laminin-511 | |
| α7 | β1 | Laminin-111, Laminin-211, Laminin-221 |
| α8 | β1 | Fibronectin, Vitronectin |
| α10 | β1 | Collagen I, Collagen IV |
| αV | β1 | Vitronectin, Fibronectin, Fibrinogen |
| β5 | Vitronectin | |
| β8 | Vitronectin, Fibronectin, Collagen IV, Laminin-111 |
- In your lab notebook, create a data table with a column called "Integrin Subunits" and list each of the integrin subunits in it (all alpha and beta subunits listed in Table 3), with one row for each subunit. Make four additional columns labeled "Expression in Adult Kidney", "Expression in 10wk Embryonic Kidney", "Expression in 16wk Embryonic Kidney", and "Expression in 20wk Embryonic Kidney". You will be filling in this data table in the following steps.
- Learn about the expression of certain integrin subunits by using NIH's Gene Database.
- Type the name of the gene you want to learn more about in the search box.
- For alpha subunits, enter "ITGA" followed by the number. For example, for α5 enter ITGA5. (For integrin αV, use ITGAV.)
- For beta subunits, enter "ITGB" followed by the number.
- This will take you to a search results webpage. Choose the "Gene" result. It is usually the top result on the page.
- On the gene's page, scroll down to the "Expression" section. This section shows you how much the gene is expressed in different tissues. Choose the "See details" option in the top right corner to take you to a page similar to Figure 2.
- For this science project, you are only interested in each subunit's expression in the kidney.
- To see the expression in adult kidney tissue, choose "HPA RNA-seq normal tissues" from the pull-down menu. The results will show in both a graph and in a table. In your data table, record the kidney RPKM values for the subunit. [Note: RPKM is a normalized way of looking at gene expression.]
- To see the expression in embryonic kidney tissue, choose "Tissue-specific circular RNA induction during human fetal development" from the pull-down menu. Record the kidney RPKM values for the subunit at 10, 16, and 20 weeks of embryonic development in your data table.
- Repeat steps 3a–3d for all for all integrin subunits in Table 3.
- Type the name of the gene you want to learn more about in the search box.
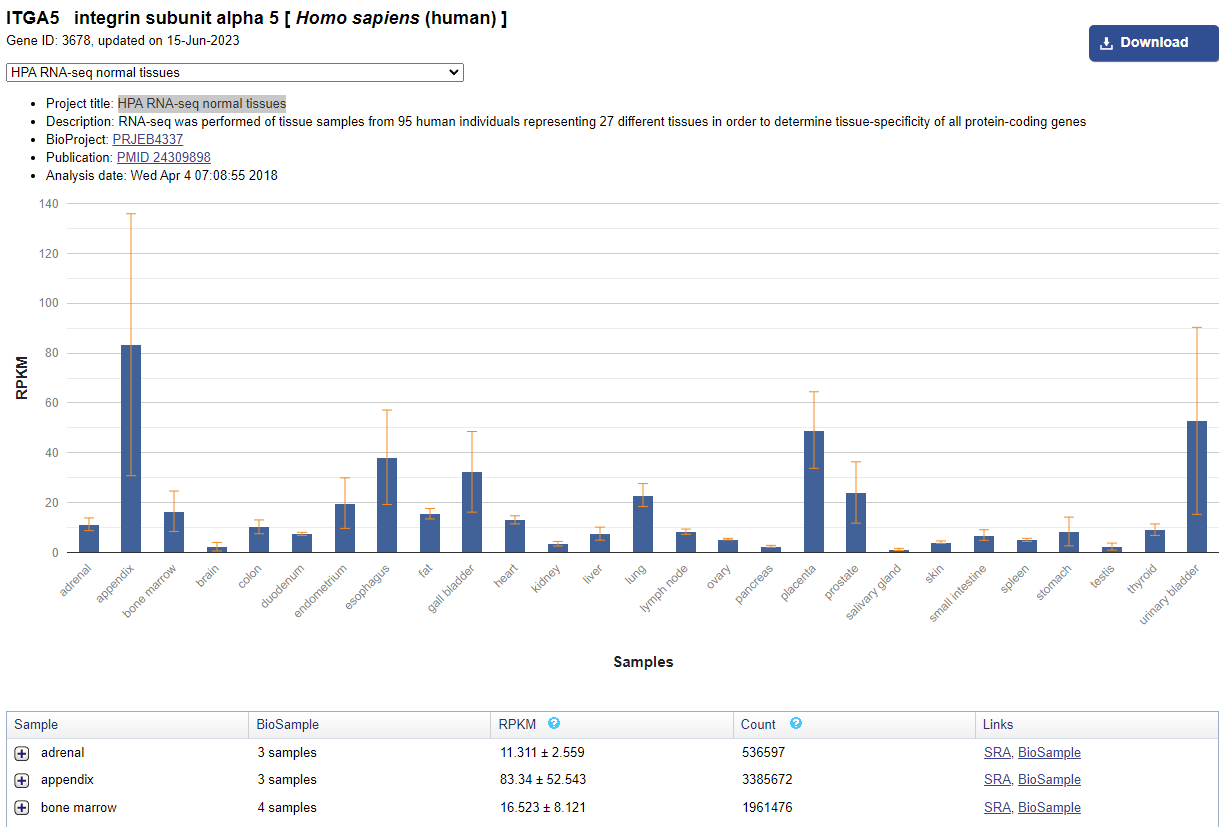 Image Credit: Public domain
Image Credit: Public domain
Figure 2. The expression data for a specific integrin subunit in NIH's Gene database will look similar to this one for the integrin α5 subunit.
- Looking at your data table, which integrin subunits are most highly expressed, or have the highest values, in the kidney cells?
- Set an expression level threshold that results in a list of several "highly expressed" integrin subunits.
- Go back to Table 3 and either print out or copy it into your lab notebook. Highlight, or mark in some other way, the alpha and beta integrin subunits that you determined to be highly expressed, and the integrin heterodimers that are highly expressed in both their alpha and beta integrin subunits.
- Which ECM proteins are bound by the highly expressed integrins? Which ECM proteins are bound by multiple highly expressed integrins? On Table 4 mark which ECM proteins are bound by at least two highly expressed integrins.
- Are the marked ECM proteins also highly expressed in the kidney during development? If so, this may make them even better candidates to use in bioengineering a kidney.
- Repeat steps 2-3 but this time use the ECM proteins you marked in step 6 instead of integrin subunits.
- Use Table 4 to help you search for the correct ECM protein names on the NIH's Gene site. Like integrins, some ECM proteins are also made up of multiple subunits that you will want to investigate.
Table 4. When searching the NIH's Gene database, use these abbreviated ECM protein names.ECM Protein(s) Name(s) to Search For Laminins - Laminins are made up of three subunits, an alpha, beta, and gamma, which are listed in order in their names in Table 4. For example, Laminin-521 is made up of an alpha 5, beta 2, and gamma 1 subunit.
- For alpha subunits, search for "LAMA" followed by the number. For example, for alpha 5 you would search for "LAMA5."
- For beta subunits, search for "LAMB" followed by the number.
- For gamma subunits, search for "LAMC" followed by the number.
Collagens - Collagens are made up of alpha subunits. For these subunits, search for "COL" followed by the number of the collagen and then the specific subunit.
- Collagen I is made up of alpha 1 and 2 subunits. For these subunits, search for "COL1A1" and "COL1A2," respectively.
- Collagen IV is made up of alpha 1, 2, 3, 4, 5, and 6 subunits. For these subunits, search for "COL4A1," COL4A2," etc.
Vitronectin Search for "VTN" Fibronectin Search for "FN1"
- In the data table you made in step 6, underline or highlight which ECM proteins are also highly expressed.
- Repeat steps 2-3 but this time use the ECM proteins you marked in step 6 instead of integrin subunits.
Creating the Right Microenvironment to Make a Kidney
Now that you have determined the ideal media conditions to test and the ideal substrates to grow the cells on, in this part of the science project you will be pulling your results together to create the overall best conditions for bioengineering a kidney, starting with a nephron.
- Based on your findings, what would you hypothesize to be the best soluble factors and ECM proteins to use for differentiating hiPSCs into certain nephron cell types?
- Review the data tables you made in "Driving Kidney Cell Differentiation Using Soluble Factors" and in "Using the Extracellular Matrix to Drive Kidney Cell Differentiation".
- Make a new data table that combines the results from these data tables. Specifically, have one column listing "Nephron Cell Type" (as listed in Table 1), one listing "Potential Combinations of Soluble Factors," and one listing "Potential ECM Proteins."
- Because the ECM proteins you selected were for the kidney as a whole, and not specific kidney cell types, you may want to test the same ECM proteins for all of the different nephron cell types. You can test ECM proteins individually or in combinations.
- What results would you expect from your experimental plan? What problems do you anticipate, and how might you be able to solve them? Why might some conditions be more efficient than others?
- Based on your findings, what do you hypothesize would be the best overall conditions in which to bioengineer a nephron or an entire kidney?
- If you could make a substrate or a kind of "skeleton" to grow the organ on, what ECM proteins should be used to coat it? Why do you think some would be better than others?
Ask an Expert
Global Connections
The United Nations Sustainable Development Goals (UNSDGs) are a blueprint to achieve a better and more sustainable future for all.
Variations
- When this project was written, scientific papers had not been published on differentiating pluripotent stem cells into Bowman's capsule cells, an essential type of nephron cell. Try doing a literature search for recent papers that have described this, investigate what soluble factors they used, and then repeat this project using your findings. For help searching the literature, read the guide to Resources for Finding and Accessing Scientific Papers. You may also find you need some help from someone experienced with stem cells to understand How to Read a Scientific Paper.
- This science project focused on bioengineering a kidney, but many other organs would make good targets for bioengineering efforts. Pick a different organ that is needed for transplants and repeat this project. To find what soluble factors researchers have tested, as shown in Table 1, you may need to search the literature, as described in the first variation.
- During development, cells that are next to each other, or neighboring cells, influence the differentiation of each other by secreting soluble factors and ECM proteins. This is why in Table 1 a study used conditioned media. (A definition of conditioned media is given immediately following Table 1.) Design a method for culturing hiPSCs with cells that would be neighboring kidney cells during development, aiming to differentiate hiPSCs into kidney cells. How is this approach similar to or different from the one used in this science project, and how do you think one would be better than the other? Could the methods be used in combination?
- ECM proteins can be used to differentiate hiPSCs, but these proteins are usually isolated from other animals, such as mice and rats. Because our bodies' immune systems may reject foreign proteins, kidneys bioengineered with proteins from mice or rats may be rejected after transplant. An alternative is to use peptides, which are short amino acid sequences in the ECM proteins that are shown to have functional activity. These short sequences can be synthesized in a laboratory, so they are not from mice or rats, and some may be able to interact with cells just as the normal-length protein does. You can learn more about ECM protein peptides and conduct a literature search, as described in the first variation, to find out what ECM peptides may work well for developing a synthetic scaffold for bioengineering a kidney. This can be based on the ECM proteins you selected in "Using the Extracellular Matrix to Drive Kidney Cell Differentiation" in step 6. ECM peptides can be specific for an ECM protein or common among many ECM proteins.
Careers
If you like this project, you might enjoy exploring these related careers:














