An Introduction to Radiation & Radiation Safety
Building your own homemade X-ray machine is not hard to do, but to do it safely requires that you learn the basics about radiation and radiation safety before beginning. This guide will give you an overview of these topics and point you to additional resources if you are interested in further reading. This guide may also be of interest to anyone generally interested in understanding radiation and radiation safety.
What is radiation?
The word "radiation" might make you think of disaster-filled science-fiction movies and mutant monsters, but the truth is radiation is a daily part of all of our lives. Radiation exists all around us. It is in our homes as part of our radios and microwave ovens, and we (like all other creatures and objects on Earth) experience extremely low levels every moment from our natural environment.
Most simply put, radiation is energy that travels through space as either waves or high speed particles. There are two types of radiation: non-ionizing radiation and ionizing radiation. Together these two types of radiation form the electromagnetic spectrum, shown below in Figure 1.
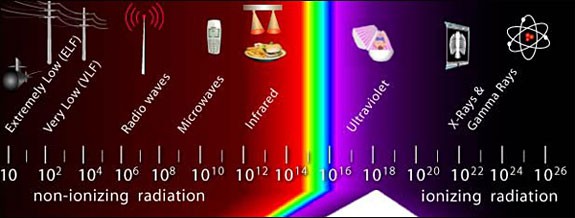 A diagram of the electromagnetic spectrum. Waves on the left side of the dagram are labeled "non-ionizing radiation" and include: Extremely Low Frequency (ELF), Very Low Frequency (VLF), Radio Waves, Microwaves, and Infrared. Visible light falls about in the middle of the spectrum. Waves on the right side of the diagram are labeled "ionizing radiation" and include: Ultraviolet, X-Rays, and Gamma Rays (highest frequency). |
| Figure 1. The electromagnetic spectrum, marked off above in various frequency (Hz) ranges, is composed of non-ionizing radiation and ionizing radiation. We spend most of our lives being constantly, and safely, exposed to many types of radiation including radio waves, microwaves, and visible light. (OSHA, 2011) |
Non-ionizing radiation does not have as much energy as ionizing radiation; thus, it is generally less harmful. The energy contained in non-ionizing radiation is enough to make the electrons in atoms or molecules wiggle and dance but not enough to strip the electrons away. In contrast, ionizing radiation has enough energy to ionize (strip away an electron leaving an unpaired electron behind) atoms and molecules, hence the name. This process and the resulting ions can cause serious damage to tissues and cells. It causes damage through several mechanisms, including:
- Breaking important chemical bonds
- Producing free radicals: these are extremely reactive ions in the body that interrupt normal function and cause cellular and tissue damage when they react with biological pathways
- Creating new, harmful, chemical bonds between macromolecules that would otherwise not occur
- Directly damaging molecules like DNA, RNA, and proteins that are responsible for normal cellular function
Depending on the amount and duration of ionizing radiation exposure, all or none of these types of damage can occur. Extremely high doses of radiation can lead to radiation sickness or even death. An above average amount of radiation exposure can be responsible for increasing one's chance of developing cancer.
Not all ionizing radiation is bad though. Ionizing radiation can be used in the medical field for imaging, for example X-rays to look at bone, or treatment as in the case of cancer radiation.
What exactly are X-rays, and how are they made?
X-rays are a type of ionizing radiation that exhibit both wave-like and particle-like properties. The wavelengths of X-rays are so short that they can travel very far through matter. For this reason they are both useful and dangerous. Safety precautions need to be taken to make sure the ionizing radiation does not damage living tissue.
X-rays are produced when electrons give up some of their energy when they react with either the nucleus of an atom or orbital electrons. There are two atomic ways for this to occur. In both cases free electrons are shot at a target of heavy atoms (like Tungsten). In the Bremsstrahlung process, the high-velocity free electrons slow down as they interact with the target atom, especially its high proton-count nucleus. This slowdown is translated into electromagnetic energy and radiated out in the form of X-rays. In the K-shell emission process, the high-velocity electrons knock out electrons from the inner orbit (K-shell) of the target atoms. Higher-energy electrons from the target atoms' outer orbit descend in to the lower-energy inner orbit to fill in the space created in the inner orbit. The descent releases excess electromagnetic energy in the form of X-rays. Tungsten is the typical target for either of these X-ray generating processes in an X-ray tube like the one shown in Figure 2 below.
 |
| Figure 2. This photograph shows what two modern X-ray tubes look like. (Raziel, Wikimedia Commons, 2007) |
Medical X-ray machines, and our homemade X-ray machine, use X-ray tubes to produce and control X-rays. These tubes have a cathode which releases electrons in to the vacuum and an anode that collects the electrons. This flow of electrons from cathode to anode is known as the beam. When the vacuum tube is hooked up to a high voltage power supply, the energy is sufficient to accelerate electrons to extreme velocities until they collide with the metal anode. This collision knocks off inner orbital electrons from the anode atoms. The subsequent act of other orbital electrons filling in the gap is what causes X-rays to be emitted. The energy of the X-rays emitted is directly proportional to the voltage applied through the tube. The minimum voltage needed to produce X-rays from a vacuum tube is approximately 20,000 V (20 kV).
How dangerous is radiation, and how is it measured?
As mentioned above in the description of "What is radiation?", ionizing radiation is capable of causing significant damage to living cells. That doesn't mean that just any exposure will cause harm. A small amount over a short period of time may be harmless. To answer whether a specific exposure is harmful or harmless, scientists focus on quantifying and measuring the dose.
There are two ways of expressing the dose. The first is the absorbed dose. This is a measure of the total amount of ionizing radiation energy absorbed by an object. The SI (International System of Units) unit for 1 joule of ionizing radiation absorbed by 1 kilogram of matter is called the gray. In the United States the absorbed dose is traditionally reported in rads as opposed to gray. Table 1 below shows how to convert between rads and gray. However, not all ionizing radiation has the same effect on human tissue. For example, alpha particles, a different type of ionizing radiation, are 20 times more likely to cause cellular damage than X-rays for an equivalent absorbed dose. To reflect those differences scientists refer to the biologically equivalent dose. To calculate the biologically equivalent dose, the absorbed dose is multiplied by a relative harm factor for that type of radiation. Sieverts are the SI unit of measurement for biologically effective dose; in the United States the traditional unit is rems. Table 1 below shows how to convert between rems and sieverts.
| Radiation Measurement | Quantifies | SI unit | Traditional unit | Equivalency |
| Absorbed dose | Conveys the raw amount of radiation energy that is absorbed by an object | gray | rad | 1 gray = 100 rad |
| Biologically effective dose | Conveys the absorbed dose corrected for how harmful that type of radiation is to human tissue | sievert | rem | 1 sievert = 100 rems |
Table 1. Radiation measurements can be quantified in a variety of ways.
When working with radiation, the biologically effective dose is what matters most for personal safety. For X-rays, the relative harm factor is 1; this means that a 1 rad absorbed dose is equivalent to a 1 rem biologically effective dose.
How much radiation am I exposed to normally, and how much is safe?
Whether it comes from the ground, the sky, or medical treatment, humans are constantly exposed to ionizing radiation from the world around them. This is a normal occurrence, and has always been the case. According to the American Nuclear Society the average person is exposed to a dose of approximately 620 mrem per year (a mrem is one thousandth of a rem). That is just an average though, and the actual figure may fluctuate widely per person depending on where they live and the medical procedures they've had that year. The international standard is to allow people who work with and around radioactive material (researchers, nuclear power plant workers, X-ray technicians etc.) to have exposures of no more than 5,000 mrem total per year. The 5,000 mrem annual dose is considered to be safe and not significantly increase the risk for radiation-related health effects.
You can estimate your own annual radiation dose using the American Nuclear Society's interactive Radiation Dose Chart.
How can I monitor my radiation exposure when experimenting with radiation?
At high biologically effective doses ionizing radiation can cause serious tissue damage. For this reason, anyone doing experiments with radioactive materials must monitor how much radiation they are being exposed to. This can be done simply with a Geiger counter. Geiger counters are tools that detect and measure ionizing radiation. Borrow or purchase a reliable Geiger counter before doing any radiation experiments!
Carefully read the instruction manual that comes with the Geiger counter, or if you are borrowing one, make sure you are carefully instructed by someone already trained to use the Geiger counter. Geiger counters typically give readings in some fraction of sieverts or rems per hour. You can use this information and the time you spend experimenting to calculate your total exposure dose (time in hours multiplied by the Geiger counter's mrem per hour reading). It is always wise to decrease your exposure as much as possible.
How can I increase my safety and diminish radiation exposure when experimenting with radiation?
When experimenting with ionizing radiation, like X-rays, diminishing your and everyone else's exposure should be the first priority. Ionizing radiation can have serious health effects at high biologically effective doses including skin burns, an increase in cancers, radiation sickness, and even death.
There are three components that figure into the total radiation dose: time, distance, and shielding.
- Time: The less time you spend exposed to radiation the lower your dose. When experimenting, make sure you know exactly what you need to get done with the radiation. If necessary, go through a dry run practicing all your motions without the radiation. Only use the radiation when you have worked out all the other potential problems with your experimental procedure.
- Distance: Like most other physical phenomena, radiation decreases with distance in accordance with the inverse square law. This means that the further away you are from the source of the radiation, the less radiation you'll be exposed to. Try to plan your experiments so that you, and anyone else around, are standing as far away from the radiation source as possible.
- Shielding: As ionizing radiation passes through matter, the intensity of the radiation is diminished. Thus, to protect yourself from radiation you should erect a barrier or shield. However, the material you use matters significantly; some materials reduce the intensity of radiation more than others. Every material has a "halving thickness." This is the thickness required to reduce the radiation intensity by half. So if the halving thickness of a material is 1 inch, then a 1 inch thick sheet will cut the radiation to 50%. Two inches will cut the radiation to 25%, 3 inches to 12.5%, and so forth. Traditionally lead is used for shielding because it has a very low halving thickness (0.4 inches). Before working with radiation, erect the shielding. When working with radiation, staying behind the shielding can be an effective way of diminishing your radiation exposure.
How much radiation can I safely be exposed to?
With radiation the answer is usually the less the better. Here are a couple of facts to keep in mind.
- A single chest X-ray or dental X-ray, both of which are considered to be medically safe, exposes a patient to 10mrem of radiation. Limiting your exposure for a single experiment to 10mrem would be similar to getting an X-ray.
- The international safety standard is 5,000mrem or less per year. Your annual exposure, both normal and experimental, should fall below the 5,000mrem level.
Where can I learn more about radiation and radiation safety?
A lot of information is available about radiation on the Internet. Here are a few reliable sources of information:
- American Nuclear Society. (2011, July 22). Radiation Dose Chart. Retrieved September 27, 2011.
- United States Center for Disease Control. (2011, March 30). Radiation and Your Health. Retrieved September 27, 2011.
- United States Environmental Protection Agency. (2010, July 19). RadTown USA. Retrieved September 27, 2011, from
- United States Nuclear Regulatory Committee. (2011, September 8). Units of Radiation Dose. Retrieved September 27, 2011.
- United States Occupational Safety and Health Administration. (2011). Radiation. Retrieved September 27, 2011.