Recycling PLA Plastic using Green Chemistry
Summary


Overview
Educational Goal
To understand:
- How polylactic acid (PLA) plastic is an example of green chemistry technology particularly pollution prevention and designing safer chemicals
Student Objectives
Students will:
- Learn about renewable "corn" plastic is made from polylactic acid
- Recycle the polylactic acid cup into a new product: a cleaning solution
- Conduct a saponification reaction
- Analyze PLA against the 12 principles of green chemistry
- (Optional) Verify the contents of their solution as lactic acid through titration
NGSS Alignment
- HS-PS1-11. Plan and conduct an investigation to compare properties and behaviors of acids and bases.
- HS-PS2-6. Communicate scientific and technical information about why the molecular-level structure is important in the functioning of the designed materials.
- HS-ETS1-3. Evaluate a solution to a complex real-world problem based on prioritized criteria and tradeoffs that account for a range of constraints including cost, safety, reliability, and aesthetics as well as potential social, cultural and environmental impacts.
- HS-ESS3-4. Evaluate or refine a technological solution that reduces impacts of human activities on natural systems.
Materials
Per student group (for PLA hydrolysis)
- Safety glasses
- Nitrile/latex gloves
- PLA cup (5g or more)
- Hot plate
- Stirring rods
- Thermometer (capable of measuring 150°C)
- 250 mL Erlenmeyer flask
- 100 mL graduated cylinder
- Watch glass
- Funnel
- Plastic pipette
- 20-25 mL of 6 M HCl
- 100 mL 1.4 M NaOH in 1:1 ethanol/water
- 1 Magnetic stir bar
- Squirt bottle or plastic spray bottles (to store lactic acid in the end)
- Scissors
- Weigh boat
- Universal pH paper
- Ice bath
Per student group (for optional Lactic Acid Titration)
- Lactic acid solution prepared by students during PLA hydrolysis
- 6 M HCl (approximately 20 mL per group)
- Graduated pipet
- Deionized water
- Titration apparatus
- 50 mL buret
- Buret clamp
- Ring stand
- Stir bar
- pH probe
- 250 mL beaker
- 0.10 M NaOH (approximately 50 mL per group)
Background Information for Teachers
The world produces approximately 200 billion pounds of plastics and nearly half of this plastic winds up in landfills each year. Polyethylene (HDPE/LDPE), polyethylene terephthalate (PET), and polystyrene (PS) are common polymers that constitute plastics, but they have enormous degradation lifetimes. These polymers can persist in the environment for 500 to 1000 years without biodegrading; current composting enzymes and bacteria are unable to break them down any faster.
Because plastics (polymers) have degradation periods significantly longer than their useful lives, they persist in landfills for enormous periods of time. Additionally, traditional plastics are produced from petroleum, which is a non-renewable resource. Taking into account the complete life cycle of plastics illustrates why there is a need for alternatives. Plastics are woven into the fabric of US society and examining alternatives to current manufacturing practices is a burgeoning area for green chemists.
Polylactic acid (PLA) is a biodegradable polymer derived from natural resources. Lactic acid, which is derived from corn, is the monomer used to create this polymer. PLA degrades under compost conditions into CO₂, H₂O, and humus, all benign components. PLA will generally breakdown under industrial compostable conditions in approximately 180 days. Because it biodegrades, the volume sent to the landfill can be reduced significantly. Also, the CO₂ released during degradation returns the carbon to the atmosphere with no overall net gain. Since the polymer is made from a corn feedstock, the process uses significantly less petroleum than traditional polymers. It is categorized under plastic resin code #7 or #0 ("other plastics"). While it is recyclable, currently there are no municipal recycling facilities that accept PLA.
Instead of disposing PLA products into landfills or compost facilities, another option of handling them at the end of the products' useful life is to reuse them as other products. One thing to do with PLA materials is to convert them into an antimicrobial cleaning solution. Figure 1 shows how PLA is made from raw materials into different products.

A flow chart of how sugarcane and corn waste are made into products made of bioplastic. The starch in sugarcane and corn waste is converted into sugars. Through fermentation, the sugars are turned into lactic acid. Oligomerization and catalytic dimerization convert the lactic acid into lactic monomers. These monomers undergo ring-opening polymerization for form polylactic acid polymer, better known as PLA plastic. The PLA plastic can then be manufactured into a variety of plastic products.
Figure 1. Conversion of corn and sugarcane waste into PLA plastic products
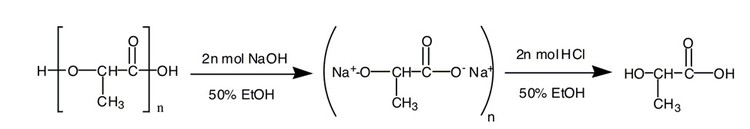
Figure 2. PLA is converted to lactic acid in a saponification reaction.
Transforming PLA to lactic acid is called a saponification reaction. The saponification reaction is an organic hydrolysis reaction that is used to make soap. (Some "green" household cleaners use lactic acid as an active ingredient). Each C-O-C bond that binds two lactic acid units together is cleaved to form 2 C-O-H units. Because the reaction occurs in a basic solution, an acidic work-up is needed to convert the deprotonated lactic acid unit (present in the form of a sodium lactate salt) into the desired protonated lactic acid form.
In an optional part two of this lesson, students can take the PLA they hydrolyzed and verify that the solution is lactic acid through titration. Since lactic acid is a weak acid, it will respond to the titration with base in a particular way.
Weak acids do not completely ionize in solution. At any given moment only some of the molecules of these acids have the acidic hydrogens released into solution. How readily a particular weak acid gives up its hydrogen is a way to characterize that substance and can be described using a number called pKa. The pKa of lactic acid is 3.86. If students' data indicates a pKa of around 3.86 it supports the assertation that the solution contains lactic acid.
For more information about Polymers and PLA check out the following sites:
- Explainer: What are Polymers? from ScienceNewsExplores
- Rezvani Ghomi E, Khosravi F, Saedi Ardahaei A, Dai Y, Neisiany RE, Foroughi F, Wu M, Das O, Ramakrishna S. The Life Cycle Assessment for Polylactic Acid (PLA) to Make It a Low-Carbon Material. Polymers (Basel). 2021 Jun 2;13(11):1854.
Teacher Preparation
For PLA hydrolysis lab
- Print student worksheets
- Locate dirty surface for students to test their cleaning solutions on. Surfaces with soap scum such as dirty classroom sinks work well as does dry spaghetti sauce on a plate.
- Prepare 1.4M NaOH in 50% ethanol in water solution. Recipe is for 1000 mL; scale as needed for the number of student groups.
- Measure 56 grams NaOH on a scale
- Transfer NaOH to 1000 mL Erlenmeyer flask
- Add 500 mL distilled water and a magnetic stir bar to the flask
- Dissolve 56 grams of NaOH in water by stirring the solution using a magnetic stirrer and the stirring hot plate
- Measure 500 mL ethanol in 500 mL graduated cylinder
- Slowly add 500 mL ethanol to flask of water and allow to mix thoroughly
- Label solution "1.4 M NaOH in 50% ethanol in water solution"
- Warning: Concentrated base can cause burns. As you are weighing out the sodium hydroxide (NaOH), wear the proper protective equipment (gloves, goggles, lab coat or apron). If the NaOH touches skin, wash immediately with copious amounts of water.
- Prepare 6 M HCl. Recipe is for 250 mL; scale as needed for the number of student groups.
- Measure 125 mL distilled water
- Add water and stir bar to Erlenmeyer flask
- Measure 125 mL 12 M HCl in graduated cylinder
- Slowly add the HCl to beaker of water
- Allow to mix thoroughly
- Dispense and label bottles "6 M HCl".
- NOTE: Always add acid to water!! This is an exothermic reaction and slowly adding the acid to water will prevent spattering of the solution.
- Warning: Concentrated acid can cause burns to skin and can pose a respiratory hazard from the fumes. When measuring and working with the hydrochloric acid, wear the appropriate protective equipment (gloves, goggles, lab coat or apron) and work in a fume hood to avoid inhalation exposure of the hydrochloric acid fumes.
For lactic acid titration lab (optional)
- Print student worksheets
- Prepare 6M HCl (same recipe as for PLA hydrolysis lab)
- Prepare 0.1 M NaOH. Recipe is for 500 ml of solution; scale as needed for the number of student groups.
- Measure 2 grams of NaOH on a scale
- Transfer to Erlenmeyer flask with a magnetic stir bar.
- Add 500mL of distilled water.
- Dissolve on stirring hot plate.
- Dispense and label "0.1 M NaOH"
- Warning: Concentrated base can cause burns. As you are weighing out the sodium hydroxide (NaOH), wear the proper protective equipment (gloves, goggles, lab coat or apron). If the NaOH touches skin, wash immediately with copious amounts of water.
Keys for Success
Each lab is designed for one class period, background reading and discussion may take additional class time. Reinforce student safety while using acids and bases. The titration lab is optional but recommended.
Before the Lab Procedure
- Assign one or more of these as student background reading. This could be done as a class or as homework reading prior to class.
- Smithsonian Magazine. Elizabeth Royte. August 2006. Accessed November 2017.
- Chemical and Engineering News. Alexander H. Tullo. Volume 95, Issue 41, p. 30-34. October 16, 2017. Accessed November 2017. 3-D printing: A tool for production Interest from manufacturers and big chemical companies shows that 3-D printing isn't just for hobbyists.
- Live Science. Mindy Weisberger. April 17, 2018. Accessed April 2018. Lab 'Accident' Becomes Mutant Enzyme That Devours Plastic.
- Chris Jordan's Gallery. Accessed August 21, 2022. Running the Numbers: Portraits of Global Mass Culture.
- Discuss the article(s) and ask students what they think about PLA.
- Explain that the goal of this lab is to recycle PLA by converting it into lactic acid, which can be used as a cleaner.
- Distribute PLA Hydrolysis Lab Procedure and PLA Hydrolysis Worksheet to each student. If using, also distribute Lactic Acid Titration Procedure and Lactic Acid Titration Worksheet to each student.
Lab Procedure
Part 1: PLA hydrolysis
- Put on safety glasses and apron/lab coat.
- Cut a PLA cup into small pieces using scissors. The smaller the pieces, the faster the reaction. Do not use any colored parts of the cup or the lip/bottom corners (thicker portions) of the cup.
- Using a weigh boat, measure 5 g of PLA pieces on a balance.
- Add the 5 g of PLA pieces to the 250 mL Erlenmeyer flask using a funnel.
- Now put on gloves!
- Using the graduated cylinder, measure 100 mL of the pre-made sodium hydroxide/ethanol solution (1.4 M NaOH in 1:1 ethanol/water) and add it to the flask.
- Add a magnetic stir bar to the flask.
- Cover the flask with a watch glass to reduce evaporation.
- Using the student lab worksheet, begin making observations about the solution.
- Place the flask onto the hot plate and heat the solution to 90°C while stirring with a stirring rod. Reduce the heat if the solution begins to vigorously boil.
- Heat and stir the solution until the PLA pieces have completely dissolved. (This usually takes 5-15 minutes).
- After the PLA pieces have completely dissolved and the solution is pale yellow, turn off the hot plate. Temperature should be 80°C-90°C.
- Use heat resistant gloves to remove the flask from the hot plate. Place the flask in an ice water bath and allow the solution to cool until it is below 60°C. This new mixture is hydrolyzed PLA.
- Using the plastic pipette, transfer 1-2 drops of the solution to the watch glass.
- Test the pH of the hydrolyzed PLA by wetting a pH strip in the watch glass. Record the pH.
- Slowly add 1-2 mL of 6 M HCl to the flask. Mix well.
- Dip the glass stirring rod into the solution then touch it to the mouth of the flask to remove big drops of liquid. The wetted tip can then be touched to a pH strip to test the solution's pH.
- Record whether the solution is acidic or basic after each test (thoroughly mix the solution after adding 6 M HCl before testing it).
- Repeat steps 16-18 until a pH of 4-5 is reached. Record the final pH. The solution now contains lactic acid and sodium chloride (NaCl).
- Using a funnel, transfer the lactic acid solution into a squirt bottle or spray bottle.
- Spray or squirt the solution onto a dirty surface and wipe clean with a paper towel.
Part 2: Lactic acid titration (optional)
- If students are unfamiliar with titration, have them review a titration tutorial before beginning.
- Remove 10 mL of the lactic acid solution from your PLA hydrolysis lab to use in the titration.
- Further acidify the 10 mL of the lactic acid solution by adding 6 M HCl until a pH of 2.5 is reached. Remember to add the hydrochloric acid slowly and mix well.
- Using a graduated pipet, add 1 mL of this solution to a clean 250 mL beaker.
- Add enough water to allow for proper pH measurements.
- Set up a titration apparatus and titrate the acid solution with the 0.10 M NaOH.
- Collect pH data after each small (1 mL or less) addition of base. Collect pH data until the pH is >11 and is constant.
- Generate a graph of the titration curve and use that to calculate pKa. If this concept is new to your students, you can share this video with them:
- Have students identify which of the 12 Principles of Green Chemistry were addressed by parts 1 and 2 of the lab.
Disposal Information
The final product from the PLA hydrolysis (the lactic acid solution) is safe to use as a cleaner but must be disposed of as laboratory waste because of high ethanol content.
Assessment
Students' lab worksheets can be collected and reviewed for comprehension. A PLA hydrolysis answer key and a lactic acid titration answer key are both available.













