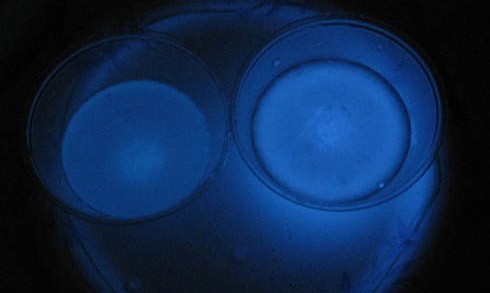
Abstract
You may have seen police investigators on TV spraying a crime scene with a liquid that glows blue if there is any blood present. Luminol is the chemical which causes the glowing. In this chemistry science fair project, you will investigate what factors make this interesting molecule "light up."Summary
Thank you to the volunteers on the Science Education Council at PPG Industries, for helpful feedback, advice and improvements on this science project.
- StyrofoamTM is a registered trademark of The Dow Chemical Company.
- Adobe® and Photoshop® are registered trademarks of Adobe Systems Incorporated in the United States and/or other countries.

Objective
In this chemistry science fair project, you will investigate how temperature affects the eerie blue glow created by the chemical luminol.
Introduction
Glow-in-the-dark objects are fascinating and mysterious. Why do they glow and where does the light come from? It seems like magic when the glow suddenly appears! There is no magic involved though, but a process called chemiluminescence. Chemiluminescent objects use chemical energy to produce light. For example, when you twist or bend a glow stick, you start a chemical reaction. One of the products of the reaction is light. The basic premise of the reaction is that the reaction between the involved chemicals releases enough energy to excite the electrons in one of the reaction partners from the ground state to the excited state, as shown in Figure 1. This causes the electrons to jump to a higher energy level and then fall back down (relax) and release light. Depending on the involved chemicals a variety of colors can be produced.
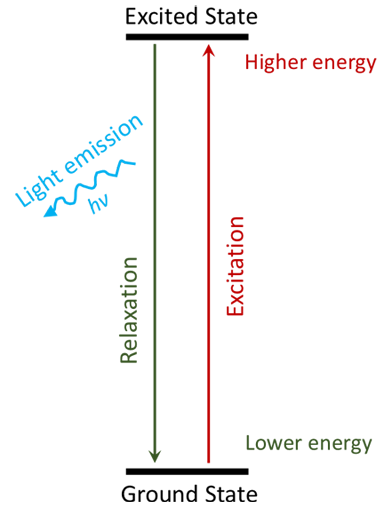
Figure 1. Simplified diagram that shows the electronic states of a molecule and the transitions between them. In a chemical reaction, electrons can be excited from the ground state to an excited state. When they relax and return to the ground state, they release their energy in the form of light.
Did you know that you can use chemiluminescence not only in glow sticks but also for solving crimes? Forensic scientists, who use science for criminal investigations, make use of chemiluminescent reactions to detect blood at crime scenes—so they can make blood visible even when it has been washed away already! When treated with a chemical called luminol, all hidden blood traces literally glow up in the dark! A demonstration of this is shown in the video below:
Luminol is a chemical that has the special property of emitting light when it reacts with certain other chemicals. The luminol reaction, as shown in Figure 2, is another example of chemiluminescence.
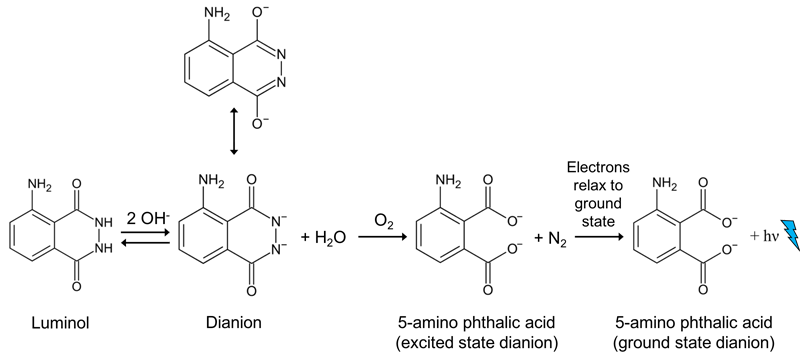
Figure 2. This is the simplified reaction mechanism for the production of light by luminol. The luminol molecule reacts with hydroxide molecules (OH-) to form the dianion (two negative charges). The dianion, which exists in two forms (the two-way arrow), reacts with oxygen to form 5-aminophthalic acid and nitrogen gas (N2). The electrons in the 5-aminophthalic acid are in an unstable, excited state. When the electrons return to their normal ground state, they release a photon of blue light.
To exhibit its luminescence, the luminol must first be activated. Usually the activator is a solution of hydrogen peroxide (H2O2) and sodium hydroxide (NaOH) in water. When luminol reacts with the hydroxide ion (OH-), a dianion is formed. A charged molecule or atom is called an ion. A molecule that has a negative charge is called an anion. And a dianion is an anion with two negative charges. The most important step in this reaction is the oxidation of luminol with oxygen, which results in the excited state dianion. The oxygen is usually formed from hydrogen peroxide (H2O2), that is added to the reaction solution. In an aqueous solution, hydrogen peroxide can decompose to produce oxygen according to the reaction below (Figure 3).
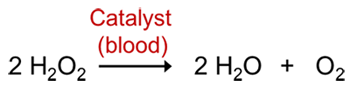
Figure 3. Decomposition reaction of hydrogen peroxide catalyzed with blood
However, this reaction proceeds very slowly. Chemical reactions can be sped up by using catalysts, which are chemicals that increase the speed of a reaction but are not actually consumed. This is where the blood comes into play! The reason why the luminol reaction makes blood glow is that it detects the iron that is present in hemoglobin, an iron-containing oxygen-transport metalloprotein in our red blood cells. This iron functions as a catalyst to speed up the production of oxygen. You can replace the hydrogen peroxide in this reaction with other oxidizing chemicals such as sodium perborate, which will release hydrogen peroxide in water according to the reaction below (Figure 4).

Figure 4. Hydrogen peroxide production from sodium perborate.
The oxygen produced from the hydrogen peroxide then reacts with the luminol dianion. The product of this reaction is very unstable and immediately decomposes with the loss of nitrogen to produce 5-aminophthalic acid. The electrons in the 5-aminophthalic acid produced by this reaction are in an excited state. Electrons in an excited state are in a higher energy level. As the excited state relaxes to the ground state, the excess energy is liberated as a photon, which is then visible as blue light.
All that a crime scene investigator has to do is to prepare a solution of luminol, sodium hydroxide (which provides the hydroxide ions for the first step of the reaction), and hydrogen peroxide and spray it throughout the area under investigation. The iron present in any blood in the area catalyzes the chemical reaction, which leads to the luminescence, revealing the location of the blood. The amount of catalyst necessary for the reaction to occur is very small, relative to the amount of luminol, allowing the detection of even trace amounts of blood. The glow lasts for about 30 seconds and is blue. Detecting the glow requires a fairly dark room. Any detected glow may be documented by a long-exposure photograph or by videotape.
In this chemistry science fair project, you will explore the luminol reaction and investigate how the solution temperature can affect the glow. In every chemical reaction, the temperature can affect how the reaction proceeds. Do you think a crime scene investigator will get the same glow from blood traces in a car during a snowy winter compared to the same car in the middle of a hot summer? You will find out in this science project with the help of a kit called the "Cool Blue Light Experiment Kit" that you can order online. Note that this kit uses perborate as the oxidizer instead of hydrogen peroxide. And don't worry, there will be no blood involved in this project— you will use copper sulfate (CuSO4) as an alternative catalyst that works just as well as real blood!
Terms and Concepts
- Chemiluminescence
- Electron
- Ground state
- Excited state
- Forensic scientist
- Luminol
- Catalyst
- (Sodium) hydroxide
- 5-aminophthalic acid
- Photon
- Hydrogen peroxide
- Ion
- Anion
- Dianion
- Oxidation
- Catalyst
- Hemoglobin
- Sodium perborate
Questions
- What other chemicals, besides iron and copper, can act as catalysts for the luminol reaction?
- Why does the light reaction stop after a certain amount of time?
- What are the roles of each of the ingredients in the "Cool Blue Light Experiment Kit"?
- How would you expect temperature to affect the amount of light produced in the luminol reaction?
- How would you expect temperature to affect how long the blue light is produced?
Bibliography
- Harris, T. (2009). How Luminol Works. Retrieved February 13, 2009.
- Helmenstine, A.M., Ph.D. (2008, March 11). Luminol Chemiluminescence Test for Blood. Retrieved February 18, 2009.
Materials and Equipment
- Cool Blue Light Experiment Kit, available from our partner Home Science Tools®.
- You will also need to gather these items, not included in the kit:
- Paper plate
- StyrofoamTM cups, 12-oz (2)
- Liquid measuring cups, 1/3-cup capacity (2)
- Kitchen thermometer
- Water
- Ice
- Metal spoons (2)
- Lab notebook
- For measuring the luminescent glow with a digital camera, you will need following materials:
- Digital camera that can take pictures in dim light (should allow for long exposures; for example, 5 sec)
- Tripod
- Stopwatch or timer
- Helper
- Optional: Image-analysis software
Disclaimer: Science Buddies participates in affiliate programs with Home Science Tools®, Amazon.com, Carolina Biological, and Jameco Electronics. Proceeds from the affiliate programs help support Science Buddies, a 501(c)(3) public charity, and keep our resources free for everyone. Our top priority is student learning. If you have any comments (positive or negative) related to purchases you've made for science projects from recommendations on our site, please let us know. Write to us at [email protected].
Experimental Procedure

Setting Up Your Materials
Note: The blue light made by the luminol reaction is best viewed in a dimly lit room. You might want to perform the experiments in a location where you can shut out surrounding light by closing the door. You will use a digital camera to capture the luminol glow.
- Read the information pamphlet that accompanies the Cool Blue Light Experiment Kit.
- When you are ready to begin investigating how temperature affects the luminol reaction, set out the two clear plastic cups that came with the kit.
- Add one scoop each of luminol and perborate mixtures to each cup as shown in Figure 5.
- Add a consistent amount of each chemical.
- Record all data in your lab notebook.
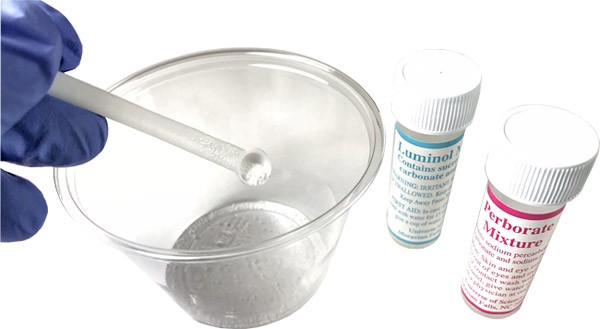
Figure 5. Preparing the luminol/perborate mixture.
- Next, add a few copper sulfate crystals to each cup.
- Try to count the grains of copper sulfate so that you are adding close to the same amount to each reaction.
- For example, try adding 10 grains of copper sulfate as shown in Figure 6. Experiment with different amounts.
- If the reaction is going to completion (that is, no more light is produced) too fast, lower the amount of copper sulfate added to the reaction.
- Try to count the grains of copper sulfate so that you are adding close to the same amount to each reaction.

Figure 6. Use a defined amount of copper sulfate crystals for each reaction.
- Note: You can also premix the dry ingredients in a clean, dry container. Add enough luminol, perborate, and copper sulfate for 10 reactions. This will minimize variation that could result if you add them separately.
- Make sure you know how to work your camera and how to set the exposure time. Set up the camera and tripod so that the camera is focused on the two cups. See Figure 7.

Figure 7. Setup for taking pictures of the luminol reaction. The digital camera is on a tripod and focusing on the two cups. The camera is set to take pictures with a 15-sec exposure time (with the lights off). The two clear plastic cups contain the chemicals for the luminol reaction. The reaction is started by adding the water from the Styrofoam cups.
- Experiment with taking pictures of the cups in dim light. For example, try 5-, 10-, and 15-sec exposures in dim light. See Figure 8. You want a picture that clearly shows the relative brightness of the two cups.
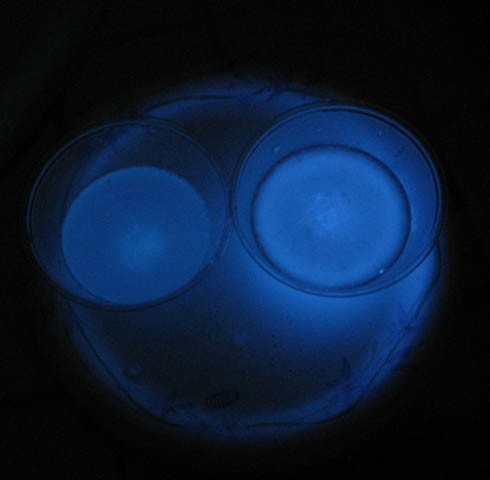
Figure 8. Two luminol reactions at different temperatures. The picture was taken with an f-stop of 8 and an exposure time 10 seconds. The reaction was started 20 seconds prior to taking the picture.
Running the Experiment
- Add 1/3 cup of ice-cold water to a Styrofoam cup.
- Add 1/3 cup of hot tap water (about 50°C) to a second Styrofoam cup.
- Determine the temperature of the water in each Styrofoam cup and record it in your lab notebook.
- Now add the cold water to one of the plastic cups containing the luminol, perborate, and copper sulfate.
- Add the hot water to the other plastic cup.
- Have your helper pour water into one of the containers so that the reactions start at the exact same time.
- You and your helper should each mix a solution with the provided popsicle sticks or a clean spoon as shown in Figure 9.

Figure 9. Once you added the water make sure to mix the solution.
- Start the stopwatch or timer.
- Dim the lights and observe the light produced by each cup.
- Take a picture of the two cups. Record the time on the stopwatch or timer at which the picture was taken.
- Continue taking pictures for about three minutes, recording the time at which each picture is taken.
- The number you take will depend on the length of the exposure.
- It is important not to vary the conditions for the pictures once you have settled on an exposure time that works well.
- A good exposure time should give you a clear picture of the two cups, so you can compare their brightness, as shown in Figure 8.
- You will want to compare all of the pictures later, so the conditions should be as consistent as possible.
- Repeat the whole experiment (steps 1–18) two more times, with clean and fresh materials.
- Be sure to use the same starting temperatures of the water.
- Take pictures at the same time intervals for each trial and using the same exposure length every time.
Analyzing Your Results
In order to graph your results, create a scale for the brightness of the light in the cups.
- Pick five pictures of the blue light in the cups that form a series from the brightest (5) to the dimmest that is still visible (1). Which reaction produced the brightest light?
- Make a figure based on these five images that shows the assigned brightness level for each number (1–5). This will be your standard for assigning a brightness level to each cup.
- Using the scale you have made, record the brightness of all of the cups, with 1 being the dimmest and 5 being the brightest.
- As an option, use an image-analysis software, such as Adobe® Photoshop®, to determine the brightness of the reactions.
- Make a data table that contains the brightness for each cup, the starting temperature, and the time at which the picture was taken.
- Convert the temperatures to Kelvin. Add 273 degrees to the temperature in Celsius to get the temperature in degrees Kelvin. Using the Kelvin scale allows you to compare the temperatures accurately.
- Average the results of the three trials for each cup.
- Graph the average brightness (1 through 5) of each reaction vs. time.
- Graph the results for the hot and the cold cups on the same chart.
- Graph the time on the x-axis and the brightness of the reaction (1–5) on the y-axis.
- Add a note on the graph indicating the starting temperature.
- How do the curves for the different starting temperatures differ in their maximum brightness and in the length of time the reaction proceeded?
Ask an Expert
Global Goals
The United Nations Sustainable Development Goals (UNSDGs) are a blueprint to achieve a better and more sustainable future for all.
Variations
- Repeat the experiment at various other starting temperatures. How does the maximum brightness vary with temperature?
- Devise a way to mix the ingredients in a way that will allow you to detect blood (from a meat tray, for example). The Cool Blue Light Experiment Kit instructions will be helpful for this.
- Experiment with a different oxidizer, such as hydrogen peroxide. How does this compare with the perborate? The Cool Blue Light Experiment Kit instructions will be helpful for this.
- If you have access to a laboratory that uses X-ray film, devise a way to record the brightness of the luminol reaction on the X-ray film.
- Try modifying the light meter in the Science Buddies science fair project What is in this Water? Experiments with a Homemade Turbidity Meter to measure the light produced by the luminol. You may need to devise a more-sensitive light detector to measure the light from the luminol reaction.
Careers
If you like this project, you might enjoy exploring these related careers: