Solar-powered Chemistry: Study Chemical Reaction Rates in Ultraviolet Beads
Abstract
Most of the ultraviolet (UV) light produced by the Sun is blocked by the atmosphere, but some UV light does still reach Earth. It can be detected using electronic devices, but can also be detected with something called UV beads. UV beads contain a pigment that changes color when they are exposed to ultraviolet radiation from the Sun. In this chemistry science fair project, you will use UV beads to study how temperature affects the rate at which they lose their color.
Summary
None
You will need to order UV beads online. See the Materials and Equipment list for details.
No issues
David B. Whyte, PhD, Science Buddies

Objective
Use UV beads to study a chemical reaction to find out how temperature affects the rate at which UV beads lose their color.
Introduction
Though ultraviolet (UV) light can be detected with electronic devices, a simple and colorful way to detect ultraviolet (UV) light is with UV-sensitive beads. UV beads change color when they are exposed to sunlight or to another ultraviolet (UV) light source, but they remain white or off-white when indoors. Because lightbulbs in homes and businesses do not produce ultraviolet light, the UV beads are colorless when kept indoors. But if you take them outside on a sunny day, UV light from the Sun turns them a variety of colors, including red, orange, yellow, blue, and purple. Some people even make bracelets from the beads so they know when they are exposed to UV light.
UV beads are made from white or clear plastic, with a photochromic dye, which means that the dye changes color when it reacts with ultraviolet light. This is a chemical reaction, which is defined as the process in which one or more substances (reactants) are chemically changed into one or more new substances (products). The ultraviolet light actually causes the shape of the molecules to change. After interacting with the UV light, the molecules change from a colorless form to a colored form. See Figure 1. The molecule undergoes a reversible rearrangement. It is reversible because the product can convert back to the reactant (this is indicated by the two arrows, one pointing left and the other pointing right). And it is a rearrangement because the parts of the molecule were rearranged.
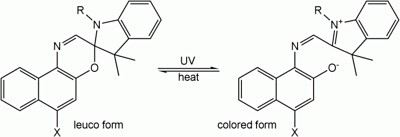
Figure 1. Example of a molecule that changes color when exposed to ultraviolet light. The colored form of the molecule is not very stable, so with just a little energy from room-temperature heat, the molecule reverts back to its colorless form. You can read the Wikipedia article on photochromism, listed in the Bibliography, for more details about this reaction. (Wikipedia, 2009.)
The goal of this chemistry science fair project is to investigate how temperature affects the speed with which the colored molecules return to their color-free state. In other words, how does temperature affect the rate at which the colored form of the molecules reverts to the color-free form? The reaction rate measures change per unit time. For this science fair project, the change will be loss of color from the beads. The time will be how long it takes, in seconds, for the change to occur.
Terms and Concepts
- Ultraviolet (UV) light
- Photochromism
- React
- Chemical reaction
- Reactant
- Product
- Molecule
- Reversible rearrangement
- Stable
- Energy
- Reaction rate
- Absolute zero
Questions
- Based on your research, what is photochromism?
- What are some other reactions that depend on light?
Bibliography
Bibliography
- Wikipedia Contributors. (2009, July 30). Photochromism. Wikipedia: The Free Encyclopedia. Retrieved September 1, 2009.
- National Aeronautics and Space Administration. (n.d.). The Electromagnetic Spectrum. Retrieved October 2, 2024.
- Chem4Kids.com. (n.d.). Rates of Reactions. Retrieved September 1, 2009.
- Helmenstine, A.M. (2019, November 26). Factors that Affect the Chemical Reaction Rate. Retrieved October 2, 2024.
- U.S. Metric Association. (n.d.). Metric System Temperatures. Retrieved October 2, 2024.
For help creating graphs, try this website:
- National Center for Education Statistics. (n.d.). Create a Graph. Retrieved June 2, 2009.
Materials and Equipment
- Foam cups, 12-ounce size (4)
- Permanent marker
- UV beads; available online from Carolina Biological, item # 956209 (1 pkg.)
- Liquid measuring cup
- Stopwatch
- Thermometer; available from Carolina Biological, item # 959876
- Lab notebook
- Microwave oven
- Graph paper
Disclaimer: Science Buddies participates in affiliate programs with Home Science Tools®, Amazon.com, Carolina Biological, and Jameco Electronics. Proceeds from the affiliate programs help support Science Buddies, a 501(c)(3) public charity, and keep our resources free for everyone. Our top priority is student learning. If you have any comments (positive or negative) related to purchases you've made for science projects from recommendations on our site, please let us know. Write to us at [email protected].
Experimental Procedure

- Perform this experiment when there is sunlight outside.
- Label the foam cups 1, 2, 3, and 4 with the permanent marker.
- Place 10 UV beads into each foam cup
- Add 250 mL of water to each cup, using the following temperatures:
- Cup # 1: Lukewarm water (20 degrees Celsius)
- Cup # 2: Ice water (0 degrees Celsius)
- Cup # 3: Lukewarm water (20 degrees Celsius)
- Cup # 4: Hot water (60 degrees Celsius)
- Take cups 2, 3, and 4 outside and expose them to the sunlight for 1 minute. Take the ice out of cup #2 before exposing the UV beads to the sunlight.
- Return indoors.
- Start the stopwatch immediately after bringing the UV beads back inside.
- Observe how long it takes the UV beads to lose their color.
- Compare the colors in the UV-exposed beads to the beads in cup # 1 (which were not exposed to UV).
- In your lab notebook, record the time at which the beads in cups 2, 3, and 4 lose their color and look like the beads in cup #1.
- Repeat steps 3–8 two more times (the beads can be reused many times). This will show that your results are reproducible.
- Graph your results.
- Convert the temperatures to kelvin (to do this, add 273 to the temperatures in degrees Celsius).
- Kelvin is a better unit to use in this science fair project because it is based on absolute zero rather than on arbitrary temperatures, such as the temperature at which water freezes. See the U.S. Metric Association website reference in the Bibliography for more information.
- Graph the temperature (kelvin) on the x-axis and the time (in seconds) on the y-axis.
- How does increasing the temperature affect how fast the UV beads lose their color?
- Convert the temperatures to kelvin (to do this, add 273 to the temperatures in degrees Celsius).
Ask an Expert
Global Goals
The United Nations Sustainable Development Goals (UNSDGs) are a blueprint to achieve a better and more sustainable future for all.
Variations
- Try more temperatures, such as 10, 30, 40, 50 degrees Celsius (convert to kelvin: 283 K, 303 K, 313 K, and 323 K).
- Experiment with different colors of the UV beads. Do UV beads of different colors have varying rates of color loss?
- Devise a way to perform the procedure at temperatures below 0 degrees Celsius.
- Based on your results, what temperature change would result in a doubling of the reaction rate? Hint: On your graph, pick two times that vary by two-fold, such as 100 seconds and 50 seconds, and determine what the temperature difference is for those two points.
- Graph 1/time on the y-axis vs. temperature on the x-axis. Because the rate measures change per unit time, the value for 1/time is proportional to the rate. Does the rate increase in a straight line as the temperature increases?
- If you want to explore this topic further, look up the Arrhenius equation. It describes mathematically how the rate of a reaction depends on the temperature.
Careers
If you like this project, you might enjoy exploring these related careers:








