Abstract
Electronic devices can be designed to detect dangerous fumes or other hazards, such as smoke or carbon monoxide. In this electronics project, you will build another potentially life-saving detector—a radon detector. Radon gas is radioactive and can pose a hazard to your health if you live in an area where it leaks from the ground. In this electronics science project, you will learn how to collect radon with an ordinary dusting cloth mounted on the intake of a fan, and then measure its radioactivity using a simple ionization chamber. The simple apparatus that you will make is full of possibilities—it can be used to track down the sources of the radioactive gas, to observe statistical equilibrium, and even to demonstrate radioactive half-life.Summary
Charles Wenzel
Edited by David B. Whyte, PhD, Science Buddies
The experimental procedure is based on material from Charles Wenzel's website (see the Bibliography), and is used with his permission.

Objective
Build a simple ionization chamber that is capable of detecting fairly low levels of radiation. You will also build a radiation collector to capture radon daughters on a dust cloth that has been mounted on the intake of a fan. This will allow you to measure the relative concentrations of radon gas in various locations and test the effectiveness of various methods of reducing the danger posed by radon daughters.
Introduction
In this electronics science project, you will build and test a device that is designed to detect radon, a radioactive gas. Before we get to the electronics, though, we should explore the basic science of radioactivity.
Atoms are found in all matter. There are stable atoms, which remain the same over billions of years, and unstable atoms, which decay into new atoms. These unstable atoms are radioactive, meaning they emit radioactivity from the nucleus as they decay. The energy that is released in the process is made up of small, fast-moving particles and high-energy waves.
Radioactivity is a random process—there is no way to know which atom in a mixture is going to decay. The radioactive substance does, however, break down at a predictable rate. The length of time for one-half of the nuclei in a radioactive sample to decay is called its half-life. A half-life can be very short (less than one millionth of a second) or very long (millions of years).
Unstable atoms, called radioisotopes (radioactive isotopes), are commonly used to study living organisms, to diagnose and treat diseases, to sterilize medical instruments and food, to produce energy for heat and electric power, and to monitor steps in various industrial processes. One kind of radioisotope that poses health risks is radon gas. Radon gas seeps from the ground and can accumulate in areas that are exposed to Earth's crust and that have inadequate ventilation. In some parts of the country, it is required by law that homeowners place radon detectors in their basements to monitor the level of radon gas. If the level of radon exceeds the established "action level," the homeowner must take steps to reduce the level of radon gas, often by increasing the ventilation.
Ordinarily, radioactive materials around you are safely bound up in solids, like trace amounts of radioactive isotopes of uranium, thorium, or radium, trapped in the concrete and earth beneath your feet. But these radioactive materials eventually decay into lighter elements, emitting energetic sub-atomic particles and radiation in the process. One of the lighter elements in the chain of decay of uranium 238 (U238) is radon, a radioactive noble gas. Since radon is a noble gas, it is chemically inert and, as a gas, it doesn't stay bound in the solid the way its parent did. It diffuses right through solids and ends up floating freely in the air. Being a noble gas, radon is fairly harmless itself. You breathe in some radon with every breath, but then you breathe it right back out, since it isn't chemically active or electrically charged. But radon has a short half-life of only about four days, meaning that about half of it will decay within four days, producing new, even lighter radioactive isotopes of other elements like polonium, lead, and bismuth. Those isotopes keep decaying, until a stable isotope of lead is reached. These radon daughters are not noble gases like radon—they are usually ionized when they are produced, and they will readily stick to anything nearby, like healthy lung tissue. They typically have an even shorter half-life than radon and quickly decay inside the lung, kicking out energetic alpha and beta particles that can cause tissue damage and potentially trigger lung cancer. This unfortunate chain of events is due to the decay chain that includes a noble gas! Radon gas is considered to be the second leading cause of lung cancer, even though her daughters are the real culprits.
This science project has three parts: 1) building a radiation detector, which is a form of ionization chamber, 2) building a radiation collector, to capture radioactive particles in the air, and 3) measuring the amount of radioactivity that is present in the air in various areas, such as in the basement of your home.
The Radiation Detector
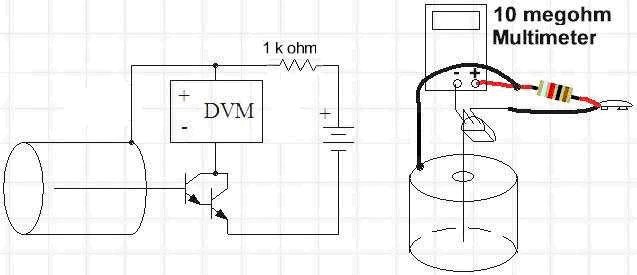
The radiation detector is based on a simple ionization chamber. An ionization chamber is simply a metal can with a wire inside. When a radioactive particle passes through the air in the chamber, many of the molecules of air are ionized, having electrons knocked loose from the outer atomic shells. Applying a positive voltage on the outer metal can, relative to the internal wire, causes the ions and electrons to move. The positive ions are attracted to the negatively charged wire, and the free electrons are attracted to the positively charged interior wall of the can. This movement of charge is a tiny current that can be amplified to detect the rate at which ions are being generated, and thereby the rate that radioactive particles are passing through the can. The simple chamber you will build is capable of detecting extremely tiny currents. 10 millivolts (mV) on the meter corresponds to 1 nanoampere (nA) flowing in the meter (10 mV / 10 megohm = 1 nA). Assuming the gain of the transistor is about 30,000, the base current is 10 nA / 30,000 = 33 femtoamperes (fA) or 0.000000000000033 amps, a very tiny current! A single femtoampere is the flow of 6,240 electrons per second, so a change of 10 mV represents a couple of hundred thousand electrons being liberated every second. There's quite a lot of activity going on in that small volume of air when you place the radioactive sample in front of the foil window and the reading climbs several tens of millivolts!
The radiation detector will be made from an empty coffee can, a single Darlington transistor, and a digital multimeter. Although its design is quite simple, it can be very sensitive, even allowing you to detect radon daughters in buildings with radon concentrations below the "action level" recommended by health authorities.
The Radiation Collector
Radon daughters will stick to just about anything they encounter, so they are easily collected by drawing air through a dusting cloth with an ordinary fan. After collecting the daughters for about an hour, the radiation being emitted from the cloth due to the further decay of the collected radioactive isotopes can be measured with the ionization chamber.
When completed, your homemade radiation detector and collector will enable you to measure the relative concentrations of radon gas in various locations and test the effectiveness of various methods of reducing the danger posed by radon daughters, a process called radon mitigation. This is an advanced electronics science project that requires a degree of independent troubleshooting, but it will demonstrate that with some basic electronic components, you can build powerful tools for measuring natural processes.
Terms and Concepts
- Radioactive
- Stable atom
- Unstable atom
- Decay
- Nucleus
- Energy
- Half-life
- Radioisotope
- Isotope
- Radon
- Uranium 238 (U238)
- Noble gas
- Chemically inert
- Polonium
- Lead
- Bismuth
- Radon daughter
- Alpha particle
- Beta particle
- Ionization chamber
- Electron
- Atomic shell
- Current
- Millivolt (mv)
- Nanoampere (nA)
- Femtoampere (fA)
- Darlington transistor
- Radon mitigation
- Amplification
- Electric field
Questions
- What are the primary sources of radon gas?
- Have buildings in your area tested positive for radon?
- Where does radon gas typically accumulate in a house?
- What elements are formed when radon decays?
- How do chemical changes in matter differ from radioactive changes? Hint: Chemistry involves changes in the electrons that surround the nuclei, and radioactivity involves changes in the nuclei.
- What kinds of fast-moving particles are produced by radioactive elements?
- How does a chemically inert gas, like radon, cause cancer?
- How does a fast-moving particle create ions in its path?
- What is meant by the term gain as applied to electronics?
- How is a Darlington transistor different than an ordinary transistor?
Bibliography
- Wikipedia Contributors. (2009, December 7). Geiger counter. Retrieved January 19, 2010.
- Centers for Disease Control and Prevention. (2013, April 5). Radiation and Your Health. Retrieved August 28, 2013.
- Environmental Protection Agency (EPA). (2009). Radon. Retrieved February 12, 2010.
- Science Buddies. (n.d.). An Introduction to Radiation & Radiation Safety. Retrieved August 28, 2013.
Please note this page within the EPA website for a map of radon levels in the U.S..
This site has additional information and figures about the radon collector and ionization chamber.
- Wenzel, C. (2009). Ion Chamber. Retrieved January 19, 2010.
Materials and Equipment
For this project, you can gather the materials yourself using this shopping list (most of the electronic components are available from Jameco Electronics).
- Large can, such as a coffee can, baby formula can, or cookie tin; about 4 inches in diameter and 5 inches tall. This large can will serve as the ionization chamber in the radon detector.
- Can opener
- Ruler
- Drill, with ¼-inch drill bit
- Safety goggles
- Tapered hand reamer; available at your local hardware store
-
MPSW45AG Darlington transistor
- Note: Other NPN Darlington transistors will work well in this science fair project, but the MPSW45AG has an extra-tall package, making it easier to keep the epoxy away from the legs. Other Darlington transistors are available from Jameco Electronics.
- 22-24 AWG Buss wire (.025 inches)
- Epoxy, Gorilla brand; available at most hardware stores
- Solder, also available from Jameco Electronics
- Soldering iron, numerous options also available from Jameco Electronics
- 1 kΩ resistor, also available from Jameco Electronics
- 9 V battery snap connector, also available from Jameco Electronics
- Assorted heat-shrink tubing, also available from Jameco Electronics
- Double-sided tape (1 roll)
- 9 V battery, also available from Jameco Electronics
- 30-AWG wrapping wire, also available from Jameco Electronics
- Short, small can, such as a small candy tin or tuna fish can; about 3 inches in diameter and 1.25 inches tall
- Fine sandpaper or a rotary tool; available at your local hardware store
- RCA phono jack connector, also available from Jameco Electronics
- Permanent marker
- Circular object with a slightly larger diameter than the large can
- Aluminum foil (1 roll)
- Scissors
- Pipe clamp
- Large rubber feet
- Digital multimeter; available at any hardware store, at Jameco Electronics, or at Amazon.com
- Squirrel cage fan
- Soup can
- Scissors, heavy-duty
- Wire strippers
- 12 V power supply, also available from Jameco Electronics
- Optional: Wire nuts or electrical tape
- Duct tape or a cable tie
- A thin dusting cloth, nylon hosiery material, or other cloth that will allow for some air flow
- Rubber band, heavy
- Lab notebook
Disclaimer: Science Buddies participates in affiliate programs with Home Science Tools®, Amazon.com, Carolina Biological, and Jameco Electronics. Proceeds from the affiliate programs help support Science Buddies, a 501(c)(3) public charity, and keep our resources free for everyone. Our top priority is student learning. If you have any comments (positive or negative) related to purchases you've made for science projects from recommendations on our site, please let us know. Write to us at [email protected].
Experimental Procedure

Important Notes Before You Begin:
Since the goal is to measure radon gas, you must have access to an area that has previously tested positive for radon with a commercial radon detection system. Testing is common in some areas of the United States, so your parents might know the levels in your own home, or you could ask friends and family. Well-ventilated homes might have few radon daughters in the air, so it's likely you will need to perform the test in an older building with lots of masonry, or in a basement. Try remote areas of the building where there is little airflow and rock-based building materials, like exposed cement or sheetrock. Basements in some parts of the country might require the meter be switched to a less-sensitive range. If you don't know of a location, this website provides general levels by state, as well as contact information for people who might be able to help you find specific locations that have already tested positive for radon: www.epa.gov/radon/zonemap.html.
Constructing the Ionization Chamber for the Radon Detector
-
Cut out one end of the large can with the can opener.
- The large can will serve as the ionization chamber (in the radon detector). The large can used could be a coffee can, baby formula can, cookie tin, or some other large can that is about 4 inches in diameter and 5 inches tall.
-
Make a 3/8-inch hole in the remaining end, as follows:
-
Find the center of the bottom of the large can.
- A simple way to find the center is to measure the diameter and divide by 2 to determine the radius.
- Now, use one of your fingernails pressed against the end of the ruler to establish the radius, and press your fingernail against the can, drawing a few lines from different directions to mark the center.
-
Find the center of the bottom of the large can.
-
It can be difficult to drill a clean hole in the thin metal, so a satisfactory alternative is to drill a smaller hole and then use a tapered hand reamer to widen the hole to about ½-inch diameter. Be sure to wear your safety goggles.
- The excess metal will form a short, ragged tube extending into the can, instead of breaking off in bits, but the hole will be smooth and round.
- Bend the base lead (middle pin) of the Darlington transistor at a right angle, extending towards the flat side of the body.
- Solder a piece of solid, 22- or 24-AWG bare wire to the base lead. See Figure 1. For more information about soldering, see the Science Buddies page How to Solder Electronic Components: Electronics Primer.

Figure 1. Darlington transistor with extended base lead.
- Cut the bare wire to be shorter than the large can, by ¾ inch.
-
Apply a small amount of epoxy to the top end of the transistor, keeping the epoxy well away from the leads.
- Caution: Don't get the epoxy on the center lead!
- Pass the long wire into the can and press the flat side of the transistor against the bottom of the can (the end with the small hole), allowing the epoxy to spread onto the can.
- You may solder a temporary wire to the can to help hold the transistor while the epoxy cures. See Figure 2. Check to make sure the center lead is free from any epoxy.

Figure 2. Epoxy applied to top of transistor. Temporary wire holds transistor until epoxy cures.
-
Solder a 1-k resistor in series with the positive (red) battery snap lead and cover the connection with heat shrink tubing or electrical tape.
- This resistor could be left out, but accidentally shorting the transistor base lead to the can would destroy the transistor. The resistor is a good idea!
- Apply some double-sided foam tape to a narrow side of a 9-V rectangular battery—the side that will result in the wires from the battery snap coming out on top.
- Stick the battery to the bottom of the ion chamber can, near the hole.
- Solder the resistor on the positive battery lead directly to the can. See Figure 3.
- Solder the negative battery lead (black) to the emitter of the transistor (left lead with the flat face down and the leads pointing away from you).
-
Connect a thin, flexible 3-in. wire to the remaining collector lead (right lead with the flat face down and the leads pointing away from you).
- This should be a thin, flexible wire so that it doesn't pull on the transistor very hard as you move things around. 30-AWG "wrapping" wire is ideal. You can see a schematic of the completed ionization chamber in Figure 4.
Building the Electronics Shielding for the Radon Detector
-
Put on your safety goggles. Drill a ¼-in. hole in the bottom of the short, small can, near the edge. The exact location is not critical.
- The short, small can should be about 3 inches in diameter and about 1.25 inches tall, as shown in Figure 5, below. It could be a small candy tin, tuna fish can, or some other similar can.
- Use fine sandpaper or a rotary tool to remove the enamel coating around the hole on the inside of the can so that the washer will make electrical contact.
-
Mount the RCA phono jack in the hole
and attach the flexible wire from the collector lead to the section of the phono jack that is on the inside of the can.
- It is hard to tighten such a connector in thin metal without damaging the connector or bending the can; so optionally, snug it up as best as you can and solder the nut to the can and threads to keep it in position.

Figure 5. The phono jack should be mounted on the small, short can, as shown here.
- Position the two cans (the large can, which will serve as the ionization chamber, and the small, short can) together, making sure the connector doesn't hit the battery and the wire stays away from the center lead of the transistor.
- Holding the two cans together, with the short can on top of the large can, mark across them with a permanent marker. See Figure 6.

Figure 6. Making a mark across the two cans and removing finish from cans for soldering. Tack soldering holds cans together and makes electrical contact.
- Sand off the finish on both cans with fine sand paper. You just need to expose the bare metal so that solder will adhere, connecting the two cans.
-
Connect the battery snap to the battery, and tack-solder the two cans together, using plenty of heat and solder.
- The circuit draws virtually no power, so the battery will last many years, essentially its shelf life.
- When soldering the cans together, let the soldering iron sit several seconds to heat both cans. Right before removing the heat, add a little more solder and the fresh flux will brighten the joint.
- Use the circular object with a slightly larger diameter than the ionization chamber can to draw a circle on a piece of aluminum foil.
- Sand the paint off the lip of the ionization chamber can so that the aluminum foil will make electrical contact.
- Use a large pipe clamp to affix the aluminum foil over the end of the can. See Figure 7.

Figure 7. Pipe clamp holds aluminum foil in place.
-
Add rubber feet to whichever side looks like the bottom. See Figure 8.
- Large, adhesive-backed rubber feet can be cut in half with heavy scissors or tin snips.
- The resulting thin feet will fit to the curved can better.

Figure 8. Rubber feet keep the chamber stable on the tabletop.
-
Connect the now-completed ionization chamber to the digital multimeter that is set to the 1- or 2-V scale. If you need help using a multimeter, check out the Science Buddies resource How to Use a Multimeter.
See Figure 9 below.
- Connect the negative lead (black) to the inside of the phono jack and connect the positive lead (red) to either can.

Figure 9. The ion chamber is connected to the multimeter as shown here.
- The reading should drop to a few tens of millivolts, depending on the room temperature. The temperature will affect the electronics, not the actual rate of radioactive decay.
Building the Radon Daughter Collection Device
- Take note of Figure 10 so you can see what the finished radon daughter collection device should look like as you go through this section.

The collector consists of a DC-powered squirrel cage fan drawing air through an empty soup can. A filter material is attached over the opposite end and secured with a rubber band.
Figure 10. Finished radon daughter collector. The collector consists of a DC-powered squirrel cage fan drawing air through an empty soup can with some filter material secured over the opposite end.
- Using the can opener, remove both ends of an ordinary soup can and remove the label:
- Affix the soup can to the intake of a squirrel cage fan using epoxy. Apply a bead of epoxy to the lip of the can, making sure to leave no gaps, and place the can on the fan's intake. See Figure 11. The can may be offset from center a little, as long as the intake is completely covered.

Figure 11. Attach the soup can to the fan with epoxy.
- After the epoxy has had plenty of time to cure, cut the connector off the end of the fan's cable with heavy-duty scissors and remove some insulation from the red and black wire with the wire strippers.
- Cut the yellow wire short. It is not needed for the fan to work.
- Cut off the end of the 12-V power supply cable and strip off some of the outer insulation.
- For the supply shown, the blue wire from the supply is positive and goes to the red wire of the fan.
- The black wire from the supply goes to the black wire of the fan.
- The other wires may be cut short. Connect the wires by twisting them together and then soldering. Cover the bare wire with electrical tape or heat-shrink tubing. See Figure 12.

Figure 12. These figures show a typical assembly using solder and heat shrink tubing. The power supply wires are connected to the fan.
- To relieve strain on the wires, wrap the cable around the fan and secure them with duct tape or a cable tie. See Figure 13. The cable tie looks neater than the duct tape, but the important point is to avoid pulling directly on the wires from the fan.

Figure 13. Wrap the cable around the can and secure it with duct tape.
- Add some feet to the bottom of the fan, as you did in the previous section's step 11, so that it can stand vertically.
- Secure a piece of the dusting cloth (or whatever you chose as a breathable material) to the open end of the can with a heavy rubber band.
- Plug in the fan and verify that it pulls air through the filter.
Testing
- Set up the radon daughter collection device in an area that is likely to have elevated radon, such as a basement.
- Turn on the fan of the collection device with the filter facing up and let it run for 1–2 hours.
- Connect the ionization chamber of the radon detector to a digital multimeter and turn on the DC voltage meter.
- Make an initial meter reading with the detector and record it in your lab notebook.
- Unplug the fan of the collection device and tip the collector over to the horizontal position.
- Move the end with the collection cloth as close to the end of the ionization chamber as possible without pressing on the foil with significant force.
-
If all is working properly, the meter reading should climb for several seconds and settle out at a significantly higher reading, indicating that radioactive material has collected on the collection cloth.
- Note: Well-ventilated homes may have few radon daughters in the air and it may be necessary to repeat the test in an older building with lots of masonry, or in a basement. Try remote areas of the building where there is little airflow and rock-based building materials like exposed cement or sheetrock. Basements in some parts of the country may require the meter be switched to a less-sensitive range.
- Clean the filter by washing it with warm soap and water.
- Run the fan in a different room or in other locations for a specific time, perhaps an hour, and measure the resulting radiation. Different locations are likely to exhibit different levels of radiation due to different levels of radon gas.
- Can you map the levels and make an educated guess as to the source of the radon? How does the radon level vary with time of day in the different locations?
Ask an Expert
Global Goals
The United Nations Sustainable Development Goals (UNSDGs) are a blueprint to achieve a better and more sustainable future for all.
Variations
- Try different collection materials to find the one that is most effective. Thicker materials, like ordinary paper towels, will collect more efficiently, but the airflow is greatly reduced. Thin materials will have greater air flow, but may let the radon daughters slip by. The best choice of filter materials might not be obvious.
- Using a timer or a clock, plot the meter reading vs. time after the radioactive filter is placed against the ionization chamber foil window. The radiation will drop exponentially as the various isotopes decay. Find the point where the meter reading has dropped halfway to the background reading and record the time. This time is the approximate average half-life of the collected isotopes.
- What is the effect of lighting a stick of incense in a room that measures a significant level of radon? Plot the effect vs. time. For continuous measurements, try pointing the filter cloth directly at the aluminum foil window, but leave a gap of about 0.2", or the thickness of a piece of corrugated cardboard. Plug in the fan and let the reading settle for an hour. The gap allows air to sneak in around the edges so you can plot the radon level continuously. The more-advanced experimenter could replace the voltmeter with a computer data taker for automatically making plots. However, there is nothing wrong with the manual labor approach!
- How hard is it to reduce the radon level in a room? Plot the radiation level before and after ventilating the room. It may take several hours for a small fan or open window to make a noticeable effect.
- Do various stone materials stored in a basement raise the radon levels? Try enclosing bags of sand, cement, plaster, etc, in a fairly airtight cardboard box, along with the collector, for a day. Turn on the fan for 1 hour to collect the radioactive isotopes and use the ionization chamber to make a measurement. Start with an empty cardboard box as a control.
- Plot the decay of all the radon in an enclosed box. Place the collector and ionization chamber in an airtight box and run the meter wire out in such a way that little air can get in or out of the box. Radon has a half-life of about four days, so the radiation should exponentially decrease as all the radon decays. After a week, the radiation level should be much lower.
- Let the collection unit remain off for a day or two. Place it near the ionization chamber, leaving the gap for air flow, as described earlier. After the meter reading has settled, turn on the fan and plot the radiation level vs. time. The reading should climb over about an hour period and eventually flatten out. This plot illustrates a process known as equilibrium. At the beginning of the plot, little radioactive material is on the filter so there is little decay occurring. But as the radiation builds up, usually at a constant rate, the amount of decay occurring on the filter increases; the more radioactive isotopes there are, the more likely there will be decays. Eventually, the increasing rate of decay equals the rate of accumulation due to the air flow. This balancing act is the equilibrium point.
Careers
If you like this project, you might enjoy exploring these related careers: