Rise to the Occasion: Investigating Requirements for Yeast Fermentation
Abstract
Did you ever wonder how yeast makes bread dough rise? This project will show you what yeast does to make this happen. You'll also investigate the conditions yeast needs to grow.Summary
Laura Tan, MedImmune
Edited by Andrew Olson, Science Buddies
Objective
The objective of this project is to detect the production of carbon dioxide and alcohol by yeast fermentation and to determine the reaction time and nutrient conditions needed for fermentation. You will learn that yeast is unicellular fungi and fermentation is the process by which yeast breaks down glucose to produce alcohol and carbon dioxide.Introduction
Yeasts are unicellular fungi. They are plantlike organisms that do not contain chlorophyll and cannot undergo photosynthesis to make their own food. Instead, yeast must get their food from their surroundings. One such food source is glucose. Complex carbohydrates, like the starch in potatoes or flour, are another food source. Yeast first break these down into simple sugar molecules, like glucose.
If oxygen is present in the yeast's environment, glucose will be broken down into carbon dioxide and water, plus energy, which the yeast can use to grow and reproduce. When oxygen is not present, yeast use an alternative pathway, called alcoholic fermentation, to extract energy from sugar. In alcoholic fermentation, glucose is broken down into alcohol, carbon dioxide and water, plus energy—but much less energy than when oxygen is present. The alcohol by-product is important for making wine and beer, while the bubbles of carbon dioxide make yeast-based dough rise.
Terms and Concepts
- Unicellular
- Alcoholic fermentation
Questions
- What is limewater? How do you make it? What happens to limewater in the presence of carbon dioxide?
Bibliography
- Home Science Tools®. (n.d.). How to Make Limewater. Retrieved June 26, 2020.
This link has pictures that show how limewater turns cloudy when exposed to carbon dioxide:
- BBS Bitesize (n.d.) Identifying products. Retrieved July 1, 2020.
This link has interesting information about yeast fermentation in bread dough:
- Corriher S (n.d.) Yeast's Crucial Roles in Breadbaking. Retrieved July 1, 2020.
Materials and Equipment
- Tall, slim glasses or clear plastic containers, such as water bottles (12)
- Bendable straws (4)
- Straight straws or stirrers (4)
- Modeling clay or play dough
- Limewater (see Bibliography)
- 1 box of glucose tablets (available at your local pharmacy; you need 6 4-gram tablets)
- Yeast (12 teaspoons total)
- Water
- White sugar
- Corn syrup
- Flour
- Vegetable oil
- Stove
- Oven gloves
- Measuring cup and spoons
- Small pot
Experimental Procedure

Part 1: The Process of Fermentation
- Make a 10% glucose solution by dissolving 6 glucose tablets (4 grams each) in 240 mL of water (about 1 cup). Gentle heating and stirring will help to dissolve the tablets. After the tablets are fully dissolved, boil the solution gently for five minutes to eliminate all oxygen in the glucose. Warning: Be careful with hot stove, pot and liquid!!
- Use oven gloves to remove the small pot of glucose from the stove. Allow it to cool (feels slightly warm to the touch).
- Add 2 teaspoon of yeast to glass/plastic container. Pour the boiled glucose solution into the same container. Use a spoon/straw to gently mix.
- Carefully pour half tablespoon of vegetable oil into the container so that it rests on top of the yeast-glucose mixture to keep oxygen from entering the system.
- Insert one bendable straw into the other bendable straw.
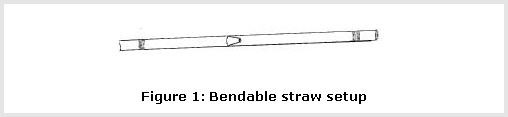
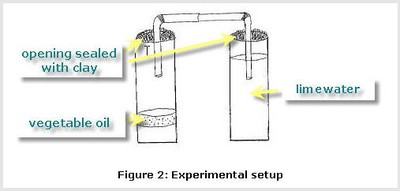
- Bend the ends of both straws at a 90-degree angle. Place the end of one straw in the mouth of the plastic/glass container you just prepared. Secure it in place with modeling clay or play dough (see Figure 2).
- Fill a second plastic/glass container about three-fourths full of limewater.
- Insert the other end of the straw set up into the mouth of plastic/glass container of limewater. The mouth of the straw should be submerged under the limewater. Secure the straw in place with modeling clay or play dough (see Figure 2).

- Leave this assembly undisturbed in a warm location for 24 hours. This condition will be called "+yeast."
- Repeat steps 1–8 but do not add the yeast. This condition will be called "−yeast." Leave it undisturbed in a warm place for 24 hours.
- After 24 hours, observe +yeast and −yeast and write down your observations in your lab notebook.
- Here are some questions to consider as you make your observations:
- What happened to the limewater for +yeast and −yeast conditions?
- The change of limewater from clear to cloudy indicates the presence of carbon dioxide. Which condition(s) produced carbon dioxide?
- Can you detect an alcohol-like odor coming from either condition?
- Production of heat suggests that a chemical reaction has occurred. Does either one feel warm?
- What is the difference between the +yeast and −yeast conditions? Why do you think this component is essential for fermentation?
Part 2: The Conditions for Fermentation
- For the second experiment, you will test four different food sources for yeast. For each condition, you will prepare two containers (three per condition is even better). Label 4 pairs of containers as follows: condition #1 water, condition #2 sugar, condition #3 flour, and condition #4 corn syrup.
- Pour 1 cup of warm water into each container.
- #1 will serve as the control with plain water. Add 1 tablespoon of sugar to each #2, 1 tablespoon of flour to each #3 and 1 tablespoon of corn syrup to each #4.
- Sprinkle 1 teaspoon of active dry yeast into all 8 containers.
- Use a clean stirrer and thoroughly stir each #1 and record the time. Repeat for all containers, using a clean stirrer for each.
- Observe the containers at regular intervals for the next 1 or 2 days. Note your observations for each container in your lab notebook. Be sure to record the time and date of each observation.
- Here are some questions to consider as you make your observations:
- Fermentation produces carbon dioxide and alcohol. Did you see evidence of either of these in each condition? Explain.
- Yeasts are one-celled, plantlike organisms that have the ability to lie dormant (inactive) if conditions are not favorable for them to grow and reproduce. Did any of the samples remain dormant? If so, which one(s)?
- What conclusion can you draw about what yeasts need?
- Which variable combined with the yeast and warm water made the yeast ferment the fastest?
Ask an Expert
Careers
If you like this project, you might enjoy exploring these related careers: