One Bad Apple Spoils the Whole Bunch: An Experiment on the Plant Hormone Ethylene
Abstract
We have all heard the old saying, "One bad apple spoils the whole bunch." Due to the production of the plant hormone ethylene during the ripening process, this saying proves true! This experiment will investigate the role of ethylene in the process of fruit ripening.
Summary
None
Require sponsor
Adult supervision required. Protective gloves, eyewear and lab coat required when working with iodine solutions.
By La Né Powers
Edited by Andrew Olson, Ph.D., Science Buddies
- This project is based on an online lab exercise by Steven K. Rice, and was used by permission:
Rice, S.K. (2001). One Bad Apple: Synchrony in Ripening Fruit. Botanical Society of America. Retrieved October 31, 2014 from https://botany.org/home/resources/plant-talking-points/one-bad-apple-synchrony-in-ripening-fruit.html
Objective
The goal of this experiment is to measure the ripening of unripe fruit induced by the plant hormone ethylene, by monitoring starch levels using an iodine solution.
Introduction
In general, plant tissues communicate using classes of compounds called hormones. These hormones are defined as substances produced in one location that have an effect on target cells in a non-adjacent location. In plants, germination, growth, development, reproduction, and environmental response are all coordinated through hormones. Although most of the main plant hormones are transported in the vascular system of the plant, one class of hormones is transferred in gaseous phase. This class includes the plant hormone ethylene.
Ethylene is manufactured and released by rapidly growing tissues (i.e., meristems) in roots, senescing flowers, and ripening fruit. For example, the darkened spots on a ripe banana release great amounts of ethylene. Ethylene has many effects on plants including being responsible for the stunting of plants in high winds or when repeatedly touched. In addition, ethylene promotes fruit ripening. Like many hormones, it does so at very low concentrations. Apple growers take advantage of this by picking fruit when it is not ripe, holding it in enclosed conditions without ethylene, and exposing it to ethylene right before taking it to market. This process is why we have newly ripened apples grown in temperate North America even in the spring and summer (apples ripen in the fall).
During the process of ripening, apples convert stored starch into sugar. In apples and many other commercial fruit, the sweet portion of the fruit evolved as a reward for animal seed dispersers. When seeds are ripe and ready for dispersal, the fruit converts stored starch, which does not taste sweet, into sugar. The hormone ethylene initiates the metabolic pathways that lead to this conversion.
Terms and Concepts
- hormone
- ethylene
Questions
- What role do plant hormones play in fruit ripening?
- What events trigger the production of ethylene?
- What physical and chemical changes occur as a fruit ripens?
- How does an iodine stain indicate the difference between ripe and unripe fruit?
Bibliography
For general information about plant hormones, including ethylene, and fruit growth and ripening, see:
- Colorado State University Extension. (December 2011). Plant Growth Factors: Plant Hormones. Retrieved October 31, 2014.
- Koning, R. E. (1994). Fruit Ripening. Plant Physiology Website, Connecticut State University. Retrieved October 31, 2014.
For an introduction to plant terms and concepts see:
- Stern, Kingsley R. 2000. Introductory Plant Biology, 8th ed. Dubuque, IA: McGraw Hill.
This project is based on an online lab exercise by Steven K. Rice, and was used by permission:
- Rice, S.K. (2001). One Bad Apple: Synchrony in Ripening Fruit. Botanical Society of America. Retrieved January 22, 2007.
Materials and Equipment
These items can be purchased from Carolina Biological Supply Company, a Science Buddies Approved Supplier:
- Protective goggles
- Chemicals for the iodine stain solution:
- 2% potassium iodide (KI) solution (455 mL)
- 2% iodine (I) solution (120 mL)
- Note: If you are ordering either of these chemicals through Carolina Biological Supply Company, the chemicals must be ordered by a teacher and shipped to a school or business address, so plan accordingly.
- Graduated cylinder, 500 mL
- Funnel
You will also need to gather these items:
- Sealable plastic bags, large enough to hold an apple and a banana (8)
- Ripe bananas (4)
- Unripe pears or unripe apples (8). Note:
- Choose apples that are in the early stages of ripening, these may be obtained at a grocery store or an orchard.
- You may also use varieties of crab apples or other local ornamental apples that are in the early stages of ripening.
- Store-bought apples may already be too ripe to conduct a ripening experiment.
- Pears are usually sold with the expectation that they will ripen after purchase, so they may be the best store-bought choice for this science project.
- Disposable gloves. Can be purchased at a local drug store or pharmacy, or through an online supplier like Carolina Biological Supply Company. If you are allergic to latex, use vinyl or polyethylene gloves.
- Lab coat. Can be purchased at scientific supply companies, or through an online supplier like Carolina Biological Supply Company. Alternatively, old, protective clothing may be used instead.
- 1 liter (or larger) brown glass or plastic bottle, with a lid, for iodine stain solution
- Shallow glass or plastic tray at least 5 cm (approx. 2") deep
- Plastic straws or glass rods
- knife and cutting board for cutting apples/pears
- Lab notebook
Disclaimer: Science Buddies participates in affiliate programs with Home Science Tools®, Amazon.com, Carolina Biological, and Jameco Electronics. Proceeds from the affiliate programs help support Science Buddies, a 501(c)(3) public charity, and keep our resources free for everyone. Our top priority is student learning. If you have any comments (positive or negative) related to purchases you've made for science projects from recommendations on our site, please let us know. Write to us at [email protected].
Experimental Procedure

- If you are unsure whether your pears (or apples) are really unripe, follow the procedure in steps 7–9 to test one before conducting your experiment.
- Label the bags:
- Bags #1–4: Control,
- Bags #5–8: Test
- In each of the Control bags, place one unripe pear (or apple) and seal the bag.
- In each of the Test bags, place one ripe banana and one unripe pear (or apple) and seal the bag.
- Place the bags together, and observe changes to fruit each day. Record your observations in your lab notebook.
- After 2–3 days, use the procedure in steps 7–9 to test the pears (or apples) for the presence of starch.
- Make the following iodine stain solution:
Safety note: Use appropriate care when handling the iodine solutions.
- Read and follow safety warnings in the Material Safety Data Sheet (MSDS) accompanying each chemical.
- Wear protective gloves, eyewear, and a lab coat when handling these chemicals.
- Do not use metal containers for storing or measuring these solutions. Iodine is corrosive to metals.
- The solutions will stain skin and clothing.
- Use the graduated cylinder to measure 455 mL of the 2% potassium iodide (KI) solution. Carefully pour into the 1 L bottle.
- Use the graduated cylinder to measure 120 mL of the 2% iodine (I) solution. Carefully pour into the 1 L bottle.
- Add water to make 1 L (about 425 mL).
- Cap bottle securely and invert several times to mix.
- The solution will keep for long periods of time in a tightly covered brown glass bottle.
- Stain the fruit:
- CAUTION: Be careful with this solution. It will stain your skin and clothing if spilled. Wear protective gloves, eyewear, and a lab coat when handling the staining solution. Read and follow the safety note (step 7).
- Pour the iodine stain solution into the glass or plastic tray to a depth of 0.5 cm.
- Cut the pear (apple) in half (in cross section). Place the cut face of the fruit into the stain. You may want to prevent the cut surface from adhering to the tray bottom by propping it using glass rods or plastic straws.
- Let the fruit soak in the stain for one minute.
- Take the fruit out and rinse the face with water. (Rinse away from the staining tray so that the rinse water does not dilute the stain.)
- Note: the stainining solution can be poured back into its storage container and used again for a future experiment. Use a funnel and pour carefully!
- Quantify the staining:
- Determine a numeric ripeness score by comparing your apples with the Ripeness Chart:
McIntosh apple starch test guide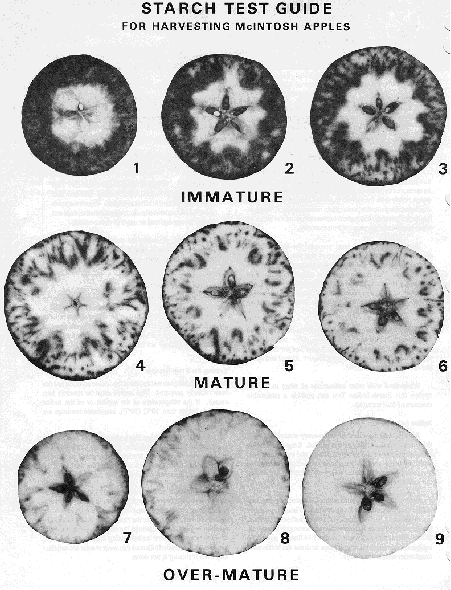
The McIntosh apple starch test guide shows nine images, 3 for each stage of immature, mature and over-mature samples. Immature samples all have a large black border surrounding the sample. Mature samples are mostly white with a small black star at the center and specks of black along the sample border. Over-mature samples are almost completely white and have blurred black stars at their center.
- Record the data in your lab notebook.
- Determine a numeric ripeness score by comparing your apples with the Ripeness Chart:
- Analyze your data:
- Summarize your data by calculating averages for Control and Test conditions.
- Compare your results in a bar graph.
Ask an Expert
Global Goals
The United Nations Sustainable Development Goals (UNSDGs) are a blueprint to achieve a better and more sustainable future for all.
Variations
- The production ethylene can also be triggered by wounds or bruises. Consider bruising the bananas in the above experiment to see if the apples will ripen more quickly.
- Do more bananas make the fruit ripen faster?
- What is the effect of temperature? Bananas in the refrigerator blacken quickly. On the other hand, refrigeration slows ripening of pears and apples. What happens if you conduct this experiment with a second set of Control and Test bags in the refrigerator?
- Ethylene is produced when fruit is removed from the parent plant. Investigate if fruit ripens more quickly on or off the plant. Tomatoes would work well for this experiment.
Careers
If you like this project, you might enjoy exploring these related careers: