Do Different Dilutions of Disinfectants Affect the Development of Bacterial Resistance?
Abstract
This is a project that illustrates the process of natural selection in action. You'll need access to a laboratory for working with the bacterial cultures used in this project.
Summary
To do this project, you will need access to a laboratory with facilities for culturing bacteria. You should be familiar with sterile technique and proper handling of bacterial cultures.
Specialty items
Standard precautions for handling bacterial cultures and bleach.
Objective
The purpose of this project is to determine if bacterial resistance to a disinfectant, after repeated exposure to a disinfectant, depends on the concentration of the disinfectant.
Introduction
Organisms evolve over generations by the process of natural selection. Individuals in every population exhibit variations in their DNA. Natural selection is the process whereby some individuals in a population are more successful at producing offspring, due to genetic variations that they possess. Over many generations, natural selection leads to an increased frequency of beneficial genes in the population.
In the laboratory, bacteria can give rise to a new generation every 15–20 minutes (Todar, 2002). This rapid generation time makes them a good experimental model for studying the effects of selection pressure on a population. In this project, you will study whether the ability of bacteria to acquire resistance to disinfectants depends on the concentration of the disinfectant.
This project has potential applications in real-life situations. When you go to the store, you can find a wide variety of antibacterial soaps available. Can left-over residue from antibacterial soaps actually work as a selection mechanism for the development of resistant bacteria?
This project uses the Kirby-Bauer disk diffusion method to measure the effectiveness of an antimicrobial agent. Here is how it works. The bacteria of interest is swabbed uniformly across a culture plate. Then a filter-paper disk, impregnated with the compound to be tested, is placed on the surface of the agar. The compound diffuses out from the filter paper into the agar. The concentration of the compound will be higher next to the disk, and will decrease gradually as distance from the disk increases. If the compound is effective against bacteria at a certain concentration, no colonies will grow wherever the concentration in the agar is greater than or equal to that effective concentration. This region is called the "zone of inhibition." Thus, the size of the zone of inhibition is a measure of the compound's effectiveness: the larger the clear area around the filter disk, the more effective the compound. Figure 1, below, illustrates the idea.
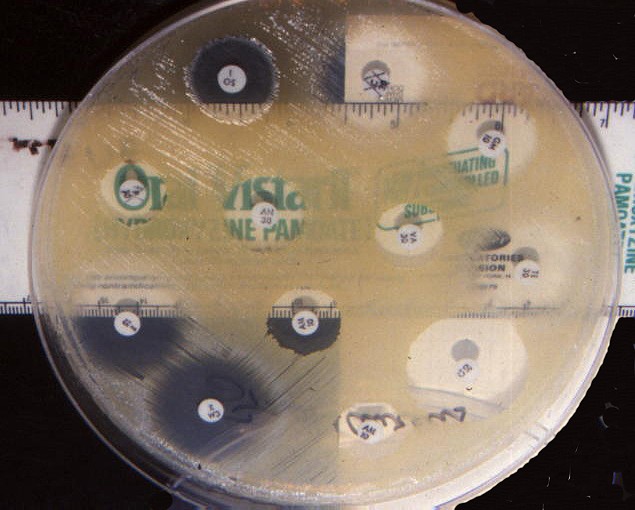
Figure 1. The illustration above shows zones of inhibition around filter paper disks saturated with anti-microbial compounds. The diameter of the zone of inhibition is a measure of the effectiveness of an anti-microbial compound (Rollins and Joseph, 2000.)
The bacteria will be subjected to five rounds of exposure, to each concentration of disinfectant. For each round, you will determine degree of resistance by measuring the zone of inhbition for each dilution of the test compound. After each round, you will select a colony closest to the disk for another round of exposure. Does resistance increase after five rounds of selection? Does the concentration of disinfectant have any effect on the development of resistance?
Terms and Concepts
To do this project, you should do research that enables you to understand the following terms and concepts:
- Colony
- Zone of inhibition
- Resistance
- Evolution
- Natural selection
Questions
- Can you think of some reasons why resistance might fail to arise in five rounds of selection?
Bibliography
- Here are references on the Kirby-Bauer disk diffusion method for measuring zones of inhibition on bacterial plates:
- Rollins, D.M. and S.W. Joseph, 2000. Antibiotic Disk Susceptibilities, Department of Cell Biology and Molecular Genetics, University of Maryland, College Park. Retrieved March 13, 2026.
- This webpage has background information on bacterial growth. It is from an online textbook of bacteriology, which can be an excellent source of further information on bacteria:
- Todar, K., 2002. Growth of Bacterial Populations, Todar's Online Textbook of Bacteriology, Department of Bacteriology, University of Wisconsin, Madison. Retrieved March 13, 2026.
- Johnson, T. and C. Case, 1995. "Chemical Methods of Control," adapted from Laboratory Experiments in Microbiology, Brief Edition, 4th ed Redwood City, CA: Benjamin/Cummings Publishing Co., available online from The National Health Museum, Access Excellence Activities Exchange. Retrieved September 11, 2006.
- Hamamah, A.A., 2004. Do Different Dilutions of Disinfectants Affect the Development of Bacterial Resistance? California State Science Fair Abstract. Retrieved September 11, 2006.
Materials and Equipment
Note: If you are carrying out this experiment in a school laboratory, which is recommended, some of the materials and equipment listed below may be more readily accessible.
These items can be purchased from Carolina Biological Supply Company, a Science Buddies Approved Supplier:
- 1.5 mL microcentrifuge tubes for making serial dilutions of disinfectants
- Sterile disks (80 per disinfectant tested, or 4 per agar plate tested). Alternatively, these could be prepared using a hole punch on filter paper.
- Sterile cotton-tipped applicators. Alternatively, cotton-tipped swabs from a new, unopened box may be used.
- Live E. coli, strain K-12.
- 20 nutrient agar plates per disinfectant to be tested:
- 4 plates per disinfectant, per round (3 dilutions + 1 control) ×
- 5 rounds of selection =
- 20 plates total per disinfectant.
- Forceps. Alternatively, tweezers may be used.
- Disposable gloves. Alternatively, these can be purchased at a local drug store or pharmacy. If you are allergic to latex, use vinyl or polyethylene gloves
You will also need to gather these items:
- Permanent marker
- 1 mL automatic pipettor with sterile tips
- Distilled or deionized water
- One or more disinfectants to test—here are some ideas for different compounds to test:
- triclosan (active ingredient in some antibacterial soaps)
- solution of garlic powder
- liquid bathroom cleaner
- liquid floor cleaner (e.g., one containing pine oil)
- mouthwash
- contact lens cleaner
- anti-acne product
- household bleach (sodium hypochlorite)
- Pencil or permanent marker
- Optional: Aluminum foil and oven
- Timer or clock
- 37°C incubator for bacterial culture plates
- Ruler, metric
- Lab notebook
Disclaimer: Science Buddies participates in affiliate programs with Home Science Tools®, Amazon.com, Carolina Biological, and Jameco Electronics. Proceeds from the affiliate programs help support Science Buddies, a 501(c)(3) public charity, and keep our resources free for everyone. Our top priority is student learning. If you have any comments (positive or negative) related to purchases you've made for science projects from recommendations on our site, please let us know. Write to us at [email protected].
Experimental Procedure

For health and safety reasons, science fairs regulate what kinds of biological materials can be used in science fair projects. You should check with your science fair's Scientific Review Committee before starting this experiment to make sure your science fair project complies with all local rules. Many science fairs follow Regeneron International Science and Engineering Fair (ISEF) regulations. For more information, visit these Science Buddies pages: Project Involving Potentially Hazardous Biological Agents and Scientific Review Committee. You can also visit the webpage ISEF Rules & Guidelines directly.
This science fair project involves the use of the bacteria E. coli. While E. coli is not considered a biohazardous or dangerous bacteria, it is important to always properly clean and dispose of bacteria and supplies that come in contact with it. See the Bacterial Safety guidelines for more details on how to handle bacterial cleanup and waste.
Preparing Plates for Disk Diffusion Test
For this experiment, it is important to innoculate the plate with a uniform distribution of bacterial colonies, and to use the exact same procedure for each plate. Here are the steps for innoculating the control and test plates.
- Note: if you want to use triclosan, here is a suggested formulation: prepare a working solution of triclosan by dissolving the powder in a solution of 17.5% ethanol and 82.5% distilled water to a final triclosan concentration of 500 μg/mL (Bittell and Hughes, 2003).
- Prepare serial two-fold dilutions of each disinfectant.
- For each disinfectant, label 3 microcentrifuge tubes with disinfectant name and tube number:
Swipe left to see moreTube # Conc. (%) 1 50% 2 25% 3 12.5% - Pipette 500 μL of water into each tube.
- Pipette 500 μL of full-strength disinfectant into the first tube. Mix thoroughly
- Using a fresh tip, pipette 500 μL of solution from the first tube into the second tube. Mix thoroughly.
- Use a fresh tip, pipette 500 μL of solution from the second tube into the third tube. Mix thoroughly.
- For each disinfectant, label 3 microcentrifuge tubes with disinfectant name and tube number:
- You can use a pencil or permanent marker to label each sterile disk with a code for the disinfectant concentration for that disk (e.g., the tube number from the serial dilution). You will need four disks for each concentration. If you label the disks, keep track of the codes in your lab notebook, wrap the disks in aluminum foil, and sterilize in a 300° oven for 30 minutes.
- Use a permanent marker to divide the bottom of the three test plates into four equal sections. Label each plate with the disinfectant and dilution to be tested.
- Use a permanent marker to divide the control plate into four equal sections. Label the plate "no disinfectant."
- Using proper sterile technique, innoculate each plate uniformly. Dip a sterile cotton-tipped applicator swab into the K-12 E. coli bacterial solution and then swab it gently across the plate. Swab in three directions (120° apart) to insure complete coverage of the plate. Cover the plate and wait at least five minutes for the plate to dry.
- For the test plates, use sterile forceps to hold a single disk by the edge with forceps and dip it into the disinfectant solution to be tested (a different concentration for each test plate). Touch the disk against the side of the tube to drain off excess liquid. Place a single disinfectant disk in the center of each of the marked sections on your test plates. Use the forceps to gently press each disk against the agar surface to insure good contact. Remember to use the exact same technique for each disk—consistency is very important for this experiment.
- For the control plate, apply sterile disks dipped in sterile water to each of the marked sections.
- Incubate all of the plates, inverted (agar on top), overnight at 37°C (or longer if necessary, for example, at lower temperature).
Measuring Zones of Inhibition
- After overnight incubation, examine your plates (keep them covered at all times).
- The control plates should show uniform colonies over the entire surface of the plate. If the distribution is highly uneven, you will need to improve your innoculation technique and repeat the experiment. The filter disks should not impede bacterial growth, since they contained only water.
- If your disinfectants are effective at the concentrations you tested, you should see zones of inhibition around the disinfectant disks. The clear zones around each disk should have a uniform width, since diffusion of the compounds through the agar should be uniform in every direction. If this is not the case, suspect either your impregnation technique, or poor contact of the filter paper with the agar.
- Measure the diameter of the zone of inhibition for each disk. Keeping the lid of the plate in place, use a ruler to measure the diameter of the disk plus the surrounding clear area in millimeters (mm).
- Include the diameter of the disk in your measurements. For example, if your disk has a diameter of 6 mm, and the clear area has a width of 3 mm beyond the disk, the diameter of the zone of inhibition that you should measure and record would be 12 mm (6 mm + 3 mm + 3 mm). This is the standard way that zones of inhibition are measured.
- You will get four separate measurements for each dilution of each disinfectant—one from each quarter section of the test plate.
- Use the values from Table 1 to evaluate the bacterial response to each compound (Johnson and Case, 1995).
Swipe left to see moreDiameter of zone of inhibition (mm) Resistant 10 or less Intermediate 11–15 Susceptible 16 or more - Are the diameters consistent across all four sections? Calculate the average and the standard deviation of the diameter of the zone of inhibition for each disinfectant.
- Do the diameters of the zones of inhibition decrease as the concentration of disinfectant decreases?
Selecting for Resistant Bacteria
Now you will select the most resistant bacteria from each plate, and repeat the exposure to diluted disinfectant. You will repeat this selection process 4 times.
- For each plate, use a sterile swab to pick up bacterial colonies growing closest to the disinfectant-impregnated disk.
- Swirl the swab in a tube of sterile water (10 ml) to dilute the bacteria. Cover the tube and agitate it.
- Repeat steps 4–9, in the Preparing Plates for Disk Diffusion Test section.
Bacterial Safety
Bacteria are all around us in our daily lives and the vast majority of them are not harmful. However, for maximum safety, all bacterial cultures should always be treated as potential hazards. This means that proper handling, cleanup, and disposal are necessary. Below are a few important safety reminders.
- Keep your nose and mouth away from tubes, pipettes, or other tools that come in contact with bacterial cultures, in order to avoid ingesting or inhaling any bacteria.
- Make sure to wash your hands thoroughly after handling bacteria.
- Proper Disposal of Bacterial Cultures
- Bacterial cultures, plates, and disposables that are used to manipulate the bacteria should be soaked in a 10% bleach solution (1 part bleach to 9 parts water) for 1–2 hours.
- Use caution when handling the bleach, as it can ruin your clothes if spilled, and any disinfectant can be harmful if splashed in your eyes.
- After bleach treatment is completed, these items can be placed in your normal household garbage.
- Cleaning Your Work Area
- At the end of your experiment, use a disinfectant, such as 70% ethanol, a 10% bleach solution, or a commercial antibacterial kitchen/bath cleaning solution, to thoroughly clean any surfaces you have used.
- Be aware of the possible hazards of disinfectants and use them carefully.
Ask an Expert
Global Goals
The United Nations Sustainable Development Goals (UNSDGs) are a blueprint to achieve a better and more sustainable future for all.
Variations
- It stands to reason that bacteria could more easily evolve resistance to antimicrobials that interfere with an organism's biochemical pathways at a single point, compared to antimicrobials that act at multiple points. Does the mechanism of action of the agent have an effect on the development of resistance? Do background research on the mechanism of action of the various disinfectants you plan to use in your project. Then design an experiment to test for development of resistance to "single-step" and "multi-step" disinfectants. Keep in mind that some bacteria have sophisticated countermeasures, such as the ability to actively pump foreign chemical compounds out of the cell. You'll also need to do background research on the mechanisms your test bacteria species use to thwart antimicrobial compounds.
- Try testing different bacterial strains. For example, you could use bacterial populations that you culture from different surfaces in your house, your skin, and your mouth.
- Do bacteria that develop resistance to lower concentrations of disinfectants survive when exposed to higher concentrations of the disinfectant? Design a continuation of this experiment to find out.
- For a more basic project on disinfectants and bacteria, see: The End Zone: Measuring Antimicrobial Effectiveness with Zones of Inhibition.
Careers
If you like this project, you might enjoy exploring these related careers: