Abstract
In this project, water fleas (Daphnia magna), a semi-transparent freshwater crustacean, are used to study the effects of caffeine on heart rate. Don't worry about having to learn how to take a crustacean's pulse: you can actually see the heart beating under a microscope. Many variations of this experiment are possible.Summary
Andrew Olson, Ph.D., Science Buddies
Sources
- The idea for this project is from:
Fitzgerald, K.F., 2003. Alcohol and Tobacco: Will It Affect Your Heart? California State Science Fair Abstract. Retrieved October 9, 2006.
Objective
The goal of this project is to test whether caffeine has an effect on heart rate, using the freshwater crustacean, Daphnia magna, as an experimental model system.
Introduction
Daphnia magna (common name "water fleas") are tiny freshwater crustaceans. They are filter feeders, and can survive in culture by eating algae, bacteria, or yeast. The body diameter of adult females is about 3–5 mm (Clare, 2002). The outer carapace of the individuals is transparent, so you can see through to the internal organs (see Figure 1, below). This allows you to monitor the heart rate of individual Daphnia that you observe with a microscope.
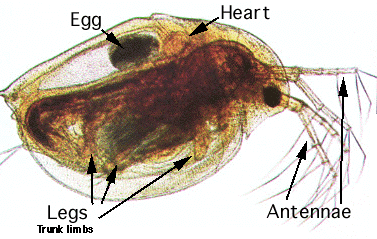 Image Credit: C.W. Jones / Copyrighted
Image Credit: C.W. Jones / CopyrightedA water flea has a heart located near the top of the head and under a small layer of skin on its back. An egg sac is located below the heart on the middle part of its back. Three legs protrude from each side of the abdoment and two antennae covered in hairs hang from the front of the head.
Figure 1. Photomicrograph of Daphnia, the common water flea. Note the position of the heart.
You can use a dissecting microscope, placing the individual Daphnia in a small drop of water so that it cannot swim out of your field of view. You can also use a compound microscope with a low-power objective. In this case, you will need a special microscope slide called a depression slide. The depression slide has a polished, concave depression at the center that can hold a large drop of water. An individual Daphnia is placed in the depression and then coverslipped for viewing. In both cases, you want to make your measurements quickly, so that the Daphnia does not become stressed in the small volume of water.
Unless you are specifically interested in the physiology of crustaceans (and there's nothing wrong with that!), the Daphnia in this experiment are what scientists call an experimental model system or simply a model system. You may be interested in how caffeine (or another drug) affects the human heart. However, designing such an experiment involving human subjects that would be both safe and scientifically valid is way beyond the level of a science fair project. That kind of experiment is for scientific and medical professionals—maybe you, later in life! A model system provides a simpler way of approaching a scientific problem.
Like any model system, Daphnia have advantages and disadvantages. On the plus side, they are fairly easy to keep, easy to study, and inexpensive. On the minus side, they are evolutionarily far-removed from humans, so you need to be cautious when interpreting your results. What happens when Daphnia are exposed to caffeine may or may not have similarities to what happens when humans drink coffee.
Before you conduct your experiment, it is a good idea to do a pilot experiment first in order to determine the right range of concentrations of caffeine to test. Ideally, your experiment will span the entire range of concentrations that produce a response (i.e., change in Daphnia heart rate). In the pilot experiment, you can test fewer individuals, and spread your test concentrations fairly widely (e.g., serial 10-fold dilutions). Once you have determined the correct concentration range to test, you will test many individuals at each concentration, and space your test concentrations more closely (e.g., serial 2-fold or 3-fold dilutions).
What kind of results do we expect to see in the pilot experiment? Let's assume that caffeine acts to increase heart rate in Daphnia. Ideally, we might hope to have data like Example 2 (red circles) in Figure 2, below. In the middle of the graph, we expect to see heart rate increasing as concentration increases. We also hope that at both extremes we will see an unchanging response. This lets us know that we've covered the both the minimum and maximum responses to the compound. If the response is still changing at either end of the graph (blue squares or green triangles), then we know we need to try at least one more concentration.
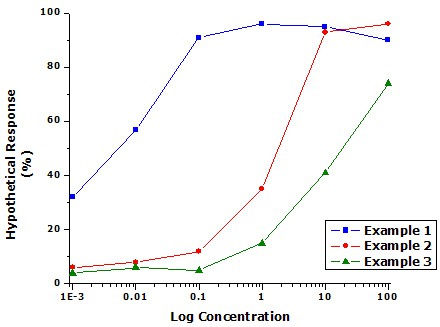
A dose response curve graph shows three separate lines with 6 data points each that simulate possible response levels based on the concentration of caffeine. The blue example 1 line has a base response level of nearly 32% for the lowest concentration of caffeine which may mean a lower concentration may need to be tested. The red and blue example lines both flatten out near the top while the green example line continues to rise until the last data point. A further data point of a higher concentration may need to be tested for the green example line.
Figure 2. Graph of simulated data illustrating possible outcomes for dose-response curves.
For the green triangles curve, at least one higher concentration should be tested. The two lowest concentrations from the pilot experiment can be excluded for the full experiment. For the blue squares curve, at least one lower concentration should be tested. The two highest concentrations from the pilot experiment can be excluded from the full experiment.
Keep in mind that is entirely possible that your curve will look different from all three of the curves in Figure 2. For example, if the test compound accelerates heart rate at intermediate concentration, but kills Daphnia at higher concentrations, heart rate will rise and then fall back to zero.
Terms and Concepts
To do this project, you should do research that enables you to understand the following terms and concepts:
- Daphnia,
- heart rate,
- caffeine.
Bibliography
- Here are two good general resources on Daphnia :
- Environmental Inquiry, 2006. "Bioassays Using Daphnia: Why Daphnia?" Cornell University and Penn State University. Retrieved October 9, 2006.
- Clare, J., 2002. Daphnia: An Aquarist's Guide, A Caudata.org website. Retrieved October 9, 2006.
- For a good start on background information on caffeine, see:
- Wikipedia contributors, 2006. Caffeine, Wikipedia, The Free Encyclopedia. Retrieved October 9, 2006.
- TOPS, 2001. Teacher Reference Pages, Caffeine Extraction Lab, Teachers+Occidental Partnership in Science, Occidental College. Retrieved August 27, 2013.
- DE/ICCA, 2002. SIDS Initial Assessment Profile, Caffeine, SIAM 14 (March): 26-28. Retrieved August 27, 2013.
- For an excellent student-written article on the effects of kava on Daphnia heart rate, see:
Kamai, J. and V. Allbrett, 2002. "Kava Decreases the Heart Rate of Daphnia," Sci-Journal. Retrieved August 27, 2013.
Materials and Equipment
These items can be purchased from Carolina Biological Supply Company, a Science Buddies Approved Supplier:
- Daphnia magna culture kit. Alternatively, you may be able to purchase Daphnia magna at a local tropical fish store.
-
Caffeine
- Alternatively, you can grind up caffeine tablets, such as sold under the brand names No-Doz and Vivarin, where the principal ingredient is 200 mg caffeine, but other ingredients are included as well.
- Note: If you purchase caffeine in powder form, you will need a digital scale with resolution down to tenths of grams, which can be purchased at Amazon.com.
- Note: If you are ordering this chemical through Carolina Biological Supply Company, the chemical must be ordered by a teacher and shipped to a school or business address, so plan accordingly.
- Springwater. Alternatively, dechlorinated tap water may be used.
- Plastic watering or transfer pipettes. You will use these to move individual Daphnia from one container to another.
- Small, fine mesh fish net. You will use this for catching and moving larger numbers of Daphnia.
- Petri dish. Alternatively, some other similar shallow container may be used.
You will also need to gather these items:
-
For observing Daphnia closely enough to monitor heart rate, you will need a microscope:
- You can use a dissecting microscope, placing the individual Daphnia to be observed in a few drops of water in a Petri dish (or similar shallow container), or
- You can also use a compound microscope with a low-magnification objective, placing the individual Daphnia to be observed in a depression slide (special microscope slide with a polished, concave depression in the center), with a cover slip.
- In either case, to avoid stressing the Daphnia in a small volume of water, make your measurements quickly, and avoid heating the solution with your illuminating light (Daphnia heart rate is temperature-sensitive).
- Small containers. These will be for holding your experimental and control groups before testing.
- Electronic timer or stopwatch
- Lab notebook
Disclaimer: Science Buddies participates in affiliate programs with Home Science Tools, Amazon.com, Carolina Biological, and Jameco Electronics. Proceeds from the affiliate programs help support Science Buddies, a 501(c)(3) public charity, and keep our resources free for everyone. Our top priority is student learning. If you have any comments (positive or negative) related to purchases you've made for science projects from recommendations on our site, please let us know. Write to us at scibuddy@sciencebuddies.org.
Experimental Procedure
For health and safety reasons, science fairs regulate what kinds of biological materials can be used in science fair projects. You should check with your science fair's Scientific Review Committee before starting this experiment to make sure your science fair project complies with all local rules. Many science fairs follow Regeneron International Science and Engineering Fair (ISEF) regulations. For more information, visit these Science Buddies pages: Project Involving Potentially Hazardous Biological Agents and Scientific Review Committee. You can also visit the webpage ISEF Rules & Guidelines directly.
Pilot Experiment to Determine Appropriate Concentration Range
- Before diving right in and trying to measure how caffeine affects Daphnia heart rate, you need to do some homework. What you would like to find out is an answer to the question, "How much caffeine (or any other compound you may be interested in studying) is a reasonable amount to try?" Once you have a good answer to this question, you can plan a pilot experiment to establish the concentration range you will be testing in your full experiment. What you would like to find, ideally, is a set of concentrations that span the entire response range. In other words, at the lowest concentration you see no resonse, and at the highest concentrations you see a maximal response, with several intermediate responses in between. The pilot experiment will help you to determine the minimum and maximum concentrations to use.
-
Below are some ideas for questions to ask when doing your background research to decide what concentrations to use in your pilot experiment:
- What amount of the compound is considered toxic?
- What is the solubility of the compound in water?
- What are typical concentrations for human consumption/use?
- A web search on "caffeine solubilty" turned up a reference (TOPS, 2001) that gives the solubility of caffeine in water as 22 milligrams/milliliter (mg/mL), which is the same as 22 grams/liter (g/L). This is the maximum amount that you could dissolve in water, and would be the absolute upper limit on the concentration range you could test.
- A web search on "caffeine lethal dose Daphnia" led to the SIAM article in the Bibliography, in the Something's Fishy About That Fertilizer, which gives the median lethal concentration (LC50) of caffeine for Daphnia as 182 mg/L of water (DE/ICCA, 2002). The LC50 is the concentration at which 50% of the organisms die when exposed to that concentration. You will obviously want your highest concentration to be less than this, because you are interested in effects on heart rate (see the Something's Fishy About That Fertilizer section for a science project using Daphnia to monitor toxicity of compounds in water). For example, you could try 100 mg/L as your highest concentration, and then do five serial 10-fold dilutions from there. Your concentrations, from highest to lowest, would then be: 100 mg/L, 10 mg/L, 1 mg/L, 0.1 mg/L, 0.01 mg/L, and 0.001 mg/L.
- Remember to use spring water to dissolve the caffeine. Tap water usually contains chlorine (and/or chloramine) as a disinfectant, which will kill Daphnia (and other aquatic organisms).
- For each caffeine concentration, measure the heart rate of at least two individual Daphnia, following the instructions in the "Measuring Daphnia Heart Rate" section, below.
- Graph your results. Since your concentration range covers 6 orders of magnitude, use a semi-log plot. Plot heart rate (y-axis) vs. log concentration (x-axis).
- Note: if you are using a compound other than caffeine for your project, and your background research does not turn up a good source for determining a starting dose, then try starting somewhere in the range of 100–200 mg/L for your highest concentration. (You want to be sure that you are looking at a specific drug effect, and not some non-specific effect such as a change in the ionic strength of the solution.)
Measuring the Effect of Caffeine at Different Concentrations
- Once you have established the concentration range over which you will run your tests, pick a set of 5–6 concentrations that span the range.
- For each of these concentrations, measure the heart rate of at least 10 individual Daphnia, following the instructions in the "Measuring Daphnia Heart Rate" section, below.
- Return the tested Daphnia to a separate container so that you don't test the same individual twice.
- Average the results for each concentration.
- Graph your results. Again, plot heart rate (y-axis) vs. log concentration (x-axis).
Measuring Daphnia Heart Rate
- Pour a small amount (10 mL) of the solution to be tested into the Petri dish.
- Use a transfer pipette to pick up an individual Daphnia, and transfer it to the Petri dish. Put the pipette under the surface of the solution in the Petri dish and pipette carefully to avoid introducing air bubbles beneath the carapace of the Daphnia. (Air bubbles make Daphnia float at the surface, unable to swim, which will eventually kill them.)
- Place the container under a dissecting microscope and focus on the Daphnia so that you can see the beating heart. If the Daphnia moves around too much, remove some of the solution in the dish.
- Count the number of heart beats that occur in 10 seconds. Use either a count-down timer with an audible signal, or have a helper time 10 seconds for you as you count heartbeats.
- Record the number of heart beats in your lab notebook. Multiply the number by 6 to get the number of beats per minute.
- Take at least three separate heart rate measurements for each individual Daphnia and calculate the average of the three measurements.
Analysis: Comparing Your Results to Human Caffeine Consumption
- More advanced students might wish to try relating Daphnia results to data for human responses to caffeine. There are many difficulties in making comparisons between data collected from different species. For example, how to account for differences in metabolism, and the vastly different body size? Do your best to consider these difficulties as you attempt to relate your results to human scale.
-
Here are some facts about effects of caffeine on humans. How do the concentrations cited below compare to your results for Daphnia
- The DE/ICCA article on caffeine describes these effects in humans: "Low doses (up to 2 mg/L in blood) stimulate the central nervous system, while high blood concentrations (10–30 mg/L) produce restlessness, excitement, tremor, tinnitus, headache, and insomnia." (DE/ICCA, 2002).
- The Wikipedia article in the Bibliography tells us that the median lethal dose (LD50, when administered at this dose, the drug is expected to be lethal to half of the recipients) of caffeine in rats is 192 mg/kg of body mass, and is thought to be between 150–200 mg/kg for humans (Wikipedia, 2006). The median lethal dose is given in mg/kg of body mass. Since blood volume is proportional to body mass, this is a rough way of normalizing the dose to blood volume. Finally, notice that the article also says, "While relatively safe for humans, caffeine is considerably more toxic to some other animals such as dogs, horses and parrots due to a much poorer ability to metabolize this compound. Caffeine has a much more significant effect on spiders, for example, than most other drugs do." (Wikipedia, 2006)
-
For caffeine, you can find typical concentrations for human consumption in the Wikipedia article cited in the Bibliography (Wikipedia, 2006).
description caffeine concentration
(mg/mL)decaffeinated coffee 0.002 typical caffeinated soda 0.096 typical drip coffee 0.056 espresso 0.096
Ask an Expert
Global Connections
The United Nations Sustainable Development Goals (UNSDGs) are a blueprint to achieve a better and more sustainable future for all.
Variations
- There are many other chemical compounds you could test, including: diphenhydramine (active ingredient in some over-the-counter decongestants), nicotine, and ethanol. As in this experiment, you should do background research and then a pilot study to determine range of concentrations appropriate to Daphnia physiology. Then prepare solutions and measure Daphnia heart rate over that range of concentrations.
- Design an experiment to study the effect of temperature on heart rate in Daphnia.
- Daphnia can also be used for toxicology studies. See the Science Buddies project Using Daphnia to Monitor Water Toxicity.
-
Advanced. The concentration of caffeine in a drink can tell you how much caffeine you would consume if you drank a known volume of liquid at that concentration. However, it does not tell you what the concentration of caffeine in your bloodstream would be. This depends on a lot of factors, e.g.:
- How well is the drug absorbed in the stomach and intestines?
- How quickly is the drug metabolized (in the stomach, liver, or elsewhere in the body)?
- What is the total volume of blood?
- From the DE/ICCA article, we can estimate one to two cups of coffee leads to a blood concentration of perhaps 2 μg/mL, and that higher doses (several cups of coffee, two cups of espresso) result in blood concentrations 5 or more times higher. (DE/ICCA, 2002). To relate effects in Daphnia to effects in humans, it would be interesting to know how caffeine concentration in the water relates to caffeine concentration inside individual Daphnia? Perhaps you can design an experiment with food coloring to see how quickly the internal fluid in Daphnia changes when the external fluid changes.
Careers
If you like this project, you might enjoy exploring these related careers:













