Eighth Grade, Chemistry Science Projects (42 results)
An experienced chemistry professor used to say that it took about one explosion per week to maintain college students' attention in chemistry lectures. At that rate, we'd get in pretty big trouble with a lot of parents and teachers! Don't worry, we still have lots of bubbles, fizzes, bangs, and color changes for you to explore.
|
Select a resource
Coding Projects
Sort by
|
Expanding gases are everywhere, from the kitchen to the cosmos. You've tasted their pleasures every time you've eaten a slice of bread, bitten into a cookie, or sipped a glass of soda. In this chemistry science fair project, you'll capture a gas in a stretchy container you're probably pretty familiar with—a balloon. This will allow you to observe the gas expansion and contraction as the temperature changes.
Read more
Have you ever had a refreshing bath using a bath bomb? A bath bomb is several ingredients mixed and molded into a shape, which becomes fizzy when it touches the water. It can be quite a relaxing experience, especially if your bath bomb has a nice fragrance or includes some bath salts. The fizz is the result of a chemical reaction taking place between different ingredients within the bath bomb. In this science project, you will get to make your own homemade bath bombs and explore how changing…
Read more
Have you ever pulled a muscle or just been sore after a long day of work or exercise? Hot showers are great, but maybe you've used the more convenient heat packs. Heat packs, which you can buy at grocery or drug stores to soothe aching muscles, use exothermic reactions to produce a low level of heat that lasts for an extended period of time. Exothermic reactions change chemical energy into heat energy. In this chemistry science fair project, you will use heat packs to study the heat-generating…
Read more
The ingredients in Alka-Seltzer® tablets undergo a chemical reaction that produces carbon dioxide gas as soon as the tablets hit water. Do you think you can cause the tablets to produce gas faster by breaking them into smaller pieces before dropping them in water? Find out for yourself with this project.
Read more
Have you ever made your own ice cream? If you have, you probably know that you need to get the ice cream mixture really cold to freeze it quickly. Ice cubes alone will not do the job, but if you add chemicals, such as salt or sugar, to the ice cubes that surround the ice cream container, the mixture gets cold enough to freeze. Why does that work? How does adding salt or sugar affect the freezing point of water? Find out with this ice-cold science project and use your results to make your own…
Read more
Instant cold packs are popular with coaches and parents for treating minor bumps and bruises. The instant cold packs are not pre-cooled—you just squeeze the cold pack and its starts to get cold. So how does it work? In this chemistry science fair project, you will investigate the chemical reaction that occurs in instant cold packs.
Read more
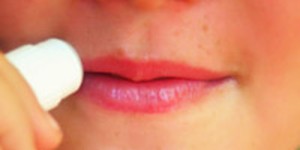
Did you know that cosmetics companies employ teams of specialized scientists to develop and test each new line of makeup, perfume, lotion, or soap? This science project lets you be the cosmetics scientist. You will create your own lip balm right in your kitchen using a short list of ingredients, then test it, and follow up with some creative cosmetics science of your own!
Read more
Have you ever tried a beauty face mask? Cosmetic face masks are skin care products that promise rapid hydration of your skin to make it look younger and more radiant. Hydrogel face masks are made of jelly-like materials that are infused with special ingredients to nurture your skin. The ingredients for cosmetic products like face masks are carefully selected by cosmetics chemists. Each product is designed to have the properties that are necessary for its intended purpose. In this science…
Read more
Many disposable diapers contain a polymer, called sodium acrylate, which is useful for absorbing water. Do background research to learn about sodium acrylate, and how it reacts with water. Make sure that you understand the terms hydrophilic, polarity, hydrogel and hydrogen bonding. Cut out the middle, padded section of a disposable diaper. Cut this into equally-sized square pieces, approximately 10-12 cm on a side. Remove some of the stuffing material so that you can staple the open…
Read more
Do you like to analyze stuff and finding out what it is made of? An analytical chemistry method, called chromatography, allows you to separate mixtures of compounds and to identify each individual compound within the mixture. Chromatography is used by many scientists, for example food scientists, forensic scientists, or organic scientists to analyze all kinds of mixtures such as food, blood, or medicine. In this project, you will be using paper chromatography to analyze the pigments from…
Read more
|